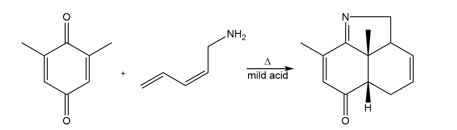
Chapter 27: Q 54. (page 1103)
Question: Draw a stepwise, detailed mechanism for the following reaction.
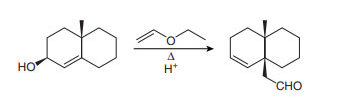
Short Answer
Answer

\
Learning Materials
Features
Discover
Chapter 27: Q 54. (page 1103)
Question: Draw a stepwise, detailed mechanism for the following reaction.

Answer

\
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What type of pericyclic reaction is illustrated in each reaction?
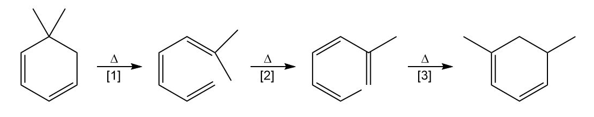
Question: Explain why heating buta-1,3-diene forms 4-vinylcyclohexene but not cycloocta-1,5-diene.
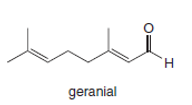
Question: What compound forms geranial (Figure 21.6) by a Cope rearrangement ?
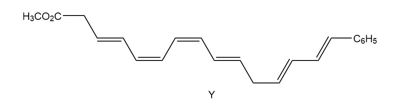
Question: The endiandric acids comprise a group of unsaturated carboxylic acids isolated from a tree that grows in the rain forests of eastern Australia. The methyl esters of endiandric acids D and E have been prepared from polyene Y by a series of two successive electrocyclic reactions: thermal ring closure of the conjugated tetraene followed by ring closure of the resulting conjugated triene. (a) Draw the structures (including stereochemistry) of the methyl esters of endiandric acids D and E. (b) The methyl ester of endiandric acid E undergoes an intramolecular [4 + 2] cycloaddition to form the methyl ester of endiandric acid A. Propose a possible structure for endiandric acid A.
Question: Draw a stepwise, detailed mechanism for the following reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.