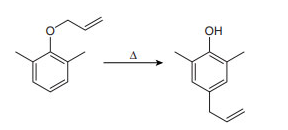
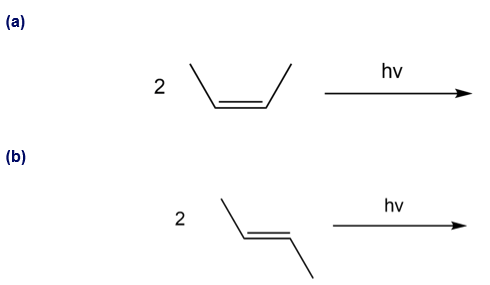
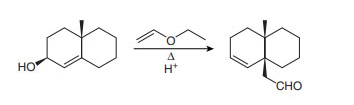
Chapter 27: Q 62. (page 1105)
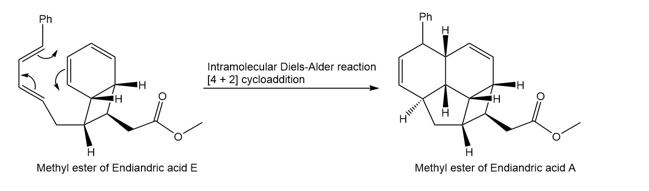
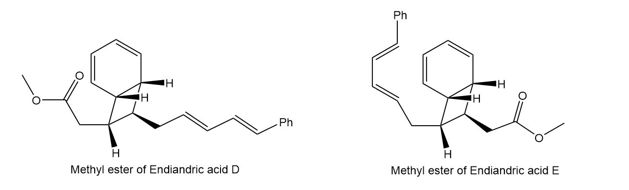
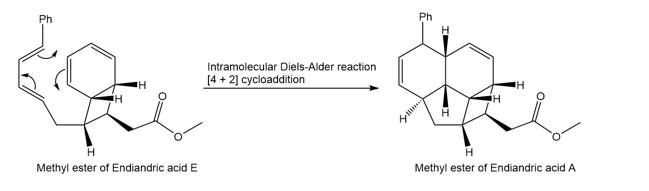
Question: The endiandric acids comprise a group of unsaturated carboxylic acids isolated from a tree that grows in the rain forests of eastern Australia. The methyl esters of endiandric acids D and E have been prepared from polyene Y by a series of two successive electrocyclic reactions: thermal ring closure of the conjugated tetraene followed by ring closure of the resulting conjugated triene. (a) Draw the structures (including stereochemistry) of the methyl esters of endiandric acids D and E. (b) The methyl ester of endiandric acid E undergoes an intramolecular [4 + 2] cycloaddition to form the methyl ester of endiandric acid A. Propose a possible structure for endiandric acid A.
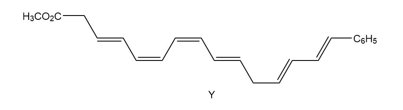
Short Answer
Answer
(a).
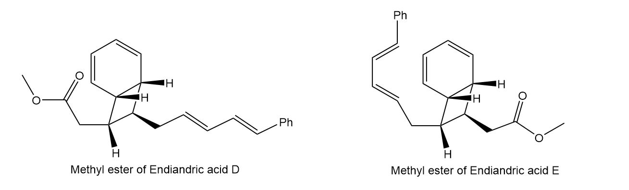
(b).