Chapter 27: Q 61. (page 1104)
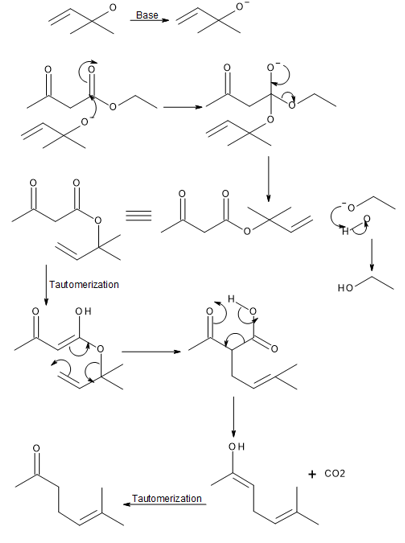
Question:Draw a stepwise mechanism for the Carroll rearrangement, a reaction that prepares aγ,δ-unsaturated carbonyl compound from aβ-keto ester and allylic alcohol in the presence ofbase.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 27: Q 61. (page 1104)
Question:Draw a stepwise mechanism for the Carroll rearrangement, a reaction that prepares aγ,δ-unsaturated carbonyl compound from aβ-keto ester and allylic alcohol in the presence ofbase.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
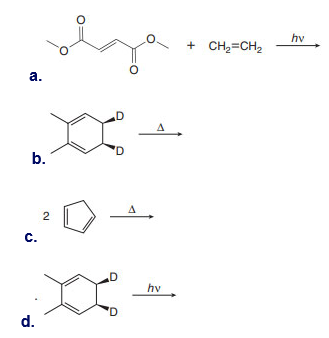
Question: Draw the product formed (including stereochemistry) in each pericyclic reaction.
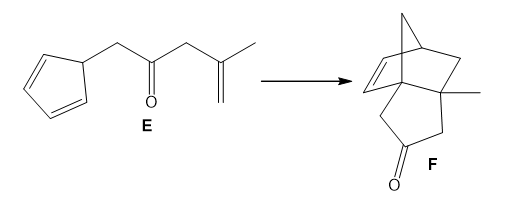
Question: Use curved arrows to show how E is converted to F by a two-step reaction sequence consisting of a [1,5] sigmatropic rearrangement followed by a [4 + 2] cycloaddition.
Question: (a) Using Figure 27.2 as a guide, draw the molecular orbitals for hexa-2,4-diene. (b) Label the HOMO and the LUMO in the ground state. (c) Label the HOMO and the LUMO in the excited state.
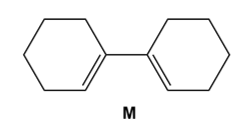
Question: Draw the product formed when diene M undergoes disrotatory cyclization. Indicate the stereochemistry at new hybridized carbons. Will the reaction occur under thermal or photochemical conditions?
Question: The bicyclic alkene P can be prepared by thermal electrocyclic ring closure from cyclodecadiene Q or by photochemical electrocyclic ring closure from cyclodecadiene R.Draw the structures of Q and R, and indicate the stereochemistry of the process by which each reaction occurs.
What do you think about this solution?
We value your feedback to improve our textbook solutions.