Chapter 27: Q 51. (page 1103)
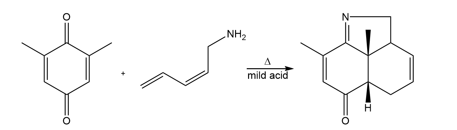
Question: Draw the product formed (including stereochemistry) in each pericyclic reaction.

Short Answer
Answer
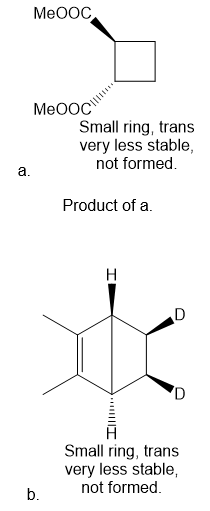
Product of b
Learning Materials
Features
Discover
Chapter 27: Q 51. (page 1103)
Question: Draw the product formed (including stereochemistry) in each pericyclic reaction.

Answer

Product of b

All the tools & learning materials you need for study success - in one app.
Get started for free
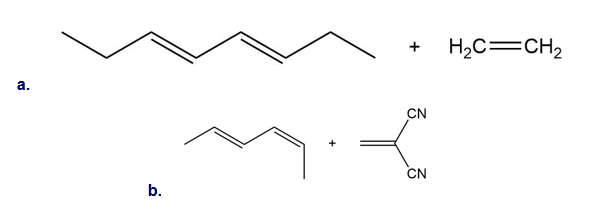
Question:Draw the product (including stereochemistry) formed from each pair ofreactants in a thermal [4 + 2] cycloaddition reaction.
Question:(a) What product is formed from the [1,7] sigmatropic rearrangement of a deuterium in the following triene?
(b) Does this reaction proceed in a suprafacial or antarafacial manner under
thermal conditions?
(c) Does this reaction proceed in a suprafacial or antarafacial manner under
photochemical conditions?

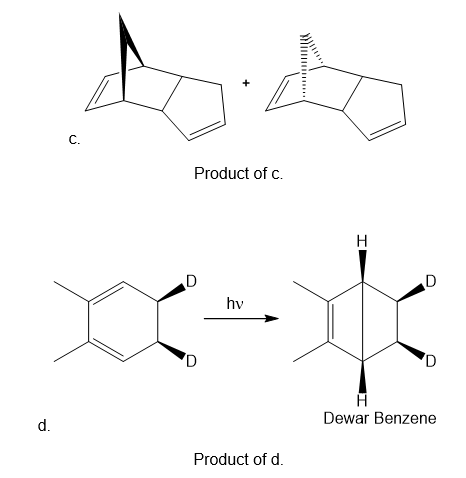
Question: What product is formed when each compound in Problem 27.6 undergoes photochemical electrocyclic ring-opening or ring closure? Label each process as conrotatory or disrotatory and clearly indicate the stereochemistry around tetrahedral stereogenic centers and double bonds.
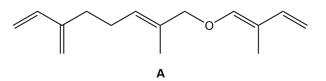
Question: Heating A results in two successive [3,3] sigmatropic rearrangements—Claisen reaction followed by Cope reaction—to afford β-Sinensal, a component of mandarin orange oil. What is the structure of β-sinensal?
Question: Draw a stepwise, detailed mechanism for the following reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.