Chapter 27: Q 14. (page 1090)
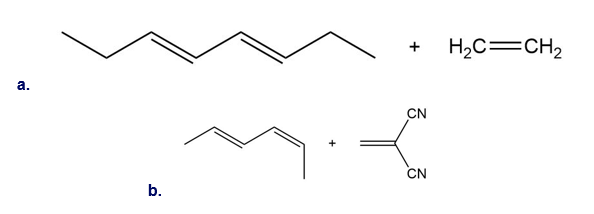
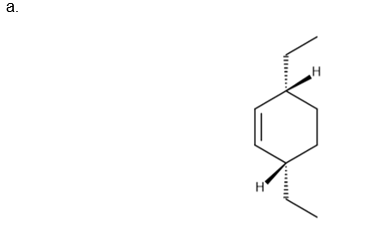
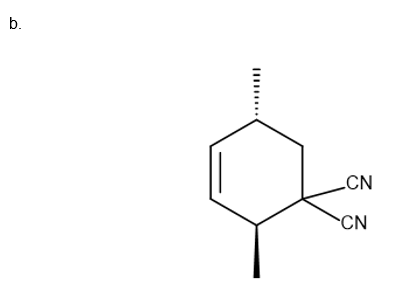
Question:Draw the product (including stereochemistry) formed from each pair ofreactants in a thermal [4 + 2] cycloaddition reaction.
Short Answer
Learning Materials
Features
Discover
Chapter 27: Q 14. (page 1090)
Question:Draw the product (including stereochemistry) formed from each pair ofreactants in a thermal [4 + 2] cycloaddition reaction.



All the tools & learning materials you need for study success - in one app.
Get started for free
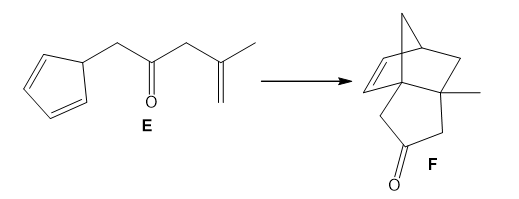
Question: Use curved arrows to show how E is converted to F by a two-step reaction sequence consisting of a [1,5] sigmatropic rearrangement followed by a [4 + 2] cycloaddition.
Question:Using the Woodward–Hoffmann rules, predict the stereochemical pathway for each cycloaddition:
(a) a [6 + 4] photochemical reaction;
(b) an [8 + 2] thermal reaction.
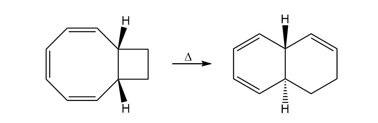
Question: Show how the following starting material is converted to the given product by a series of two pericyclic reactions. Account for the observed stereochemistry.
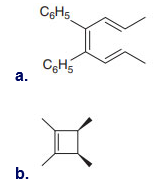
Question: What product is formed when each compound undergoes thermal electrocyclic ring-opening or ring closure? Label each process as conrotatory or disrotatory and clearly indicate the stereochemistry around tetrahedral stereogenic centers and double bonds.
Question: A solution of 5-methylcyclopenta-1,3-diene rearranges at room temperature to a mixture containing 1-methyl-, 2-methyl-, and 5-methylcyclopenta-1,3-diene. (a) Show how both isomeric products are formed from the starting material by a sigmatropic rearrangement involving a C–H bond. (b) Explain why 2-methylcyclopenta-1,3-diene is not formed directly from 5-methylcyclopenta-1,3-diene by a [1,3] rearrangement.
What do you think about this solution?
We value your feedback to improve our textbook solutions.