Chapter 27: Q 50. (page 1103)
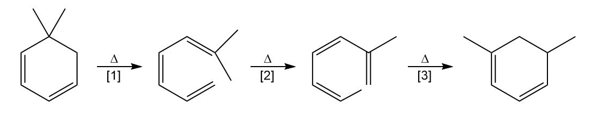
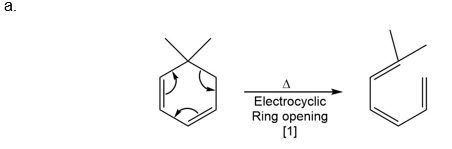
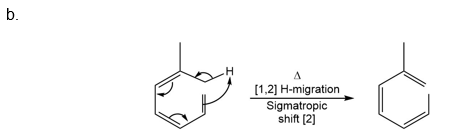
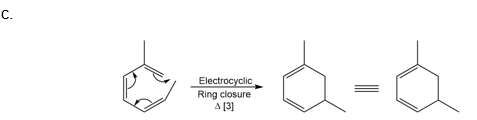
Question: What type of pericyclic reaction is illustrated in each reaction?
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 27: Q 50. (page 1103)
Question: What type of pericyclic reaction is illustrated in each reaction?

Answer



All the tools & learning materials you need for study success - in one app.
Get started for free
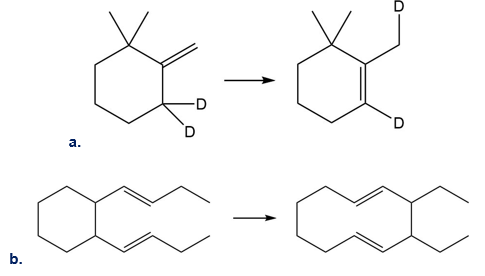
Question:What type of sigmatropic rearrangement is illustrated in each equation?
Question: (a) Using Figure 27.2 as a guide, draw the molecular orbitals for hexa-2,4-diene. (b) Label the HOMO and the LUMO in the ground state. (c) Label the HOMO and the LUMO in the excited state.
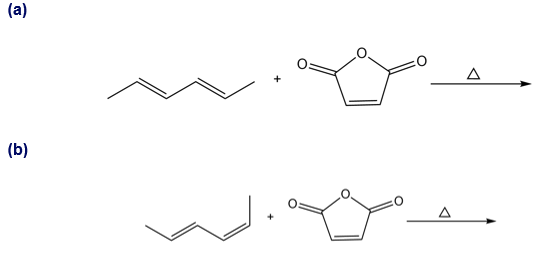
Question: Draw the product of each Diels–Alder reaction and indicate the stereochemistry at all stereogenic centers
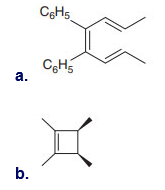
Question: What product is formed when each compound undergoes thermal electrocyclic ring-opening or ring closure? Label each process as conrotatory or disrotatory and clearly indicate the stereochemistry around tetrahedral stereogenic centers and double bonds.
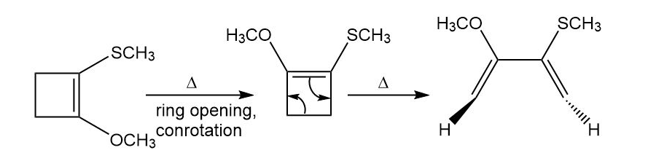
Question: Show how the following starting material is converted to the given product by a series of two pericyclic reactions. Account for the observed stereochemistry.
What do you think about this solution?
We value your feedback to improve our textbook solutions.