Chapter 27: Q 60. (page 1104)
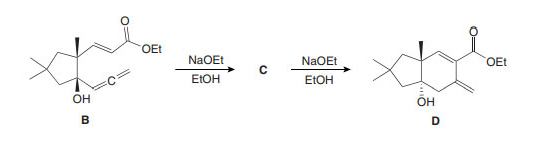
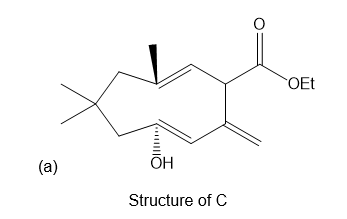
Question: a) What is the structure of C, which is formed by oxy-Cope rearrangement of B with NaOEt?
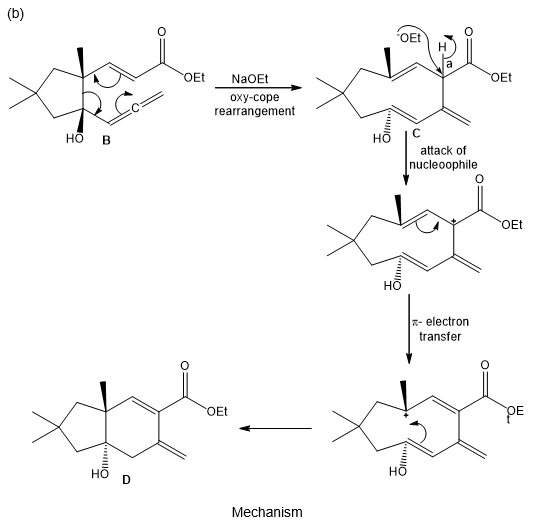
(b) Draw a stepwise mechanism for the conversion of C to the bicyclic alcohol D.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 27: Q 60. (page 1104)
Question: a) What is the structure of C, which is formed by oxy-Cope rearrangement of B with NaOEt?
(b) Draw a stepwise mechanism for the conversion of C to the bicyclic alcohol D.

Answer


All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Show how the following starting material is converted to the given product by a series of two pericyclic reactions. Account for the observed stereochemistry.
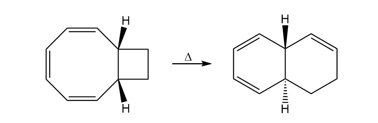
Question: One step in the synthesis of periplanone B, the chapter-opening molecule, involved anionic oxy-Cope rearrangement of the following unsaturated alcohol. Draw the product that results after protonation of the intermediate enolate.
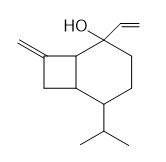
Question: Draw a stepwise, detailed mechanism for the following reaction.
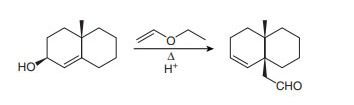
Question: What product is formed from the Cope or oxy-Cope rearrangement of each starting material?
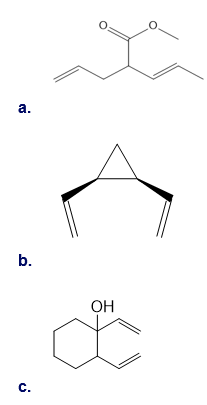
Question: Draw the product formed (including stereochemistry) in each pericyclic reaction.
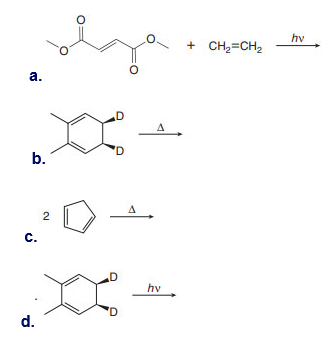
What do you think about this solution?
We value your feedback to improve our textbook solutions.