Chapter 27: Q 42. (page 1102)
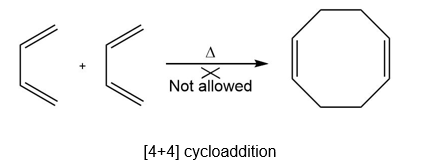
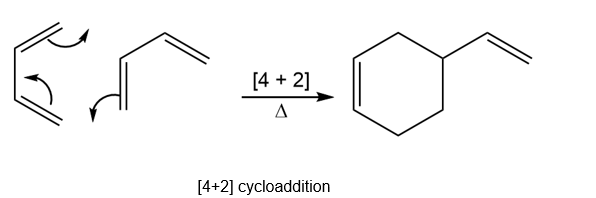
Question: Explain why heating buta-1,3-diene forms 4-vinylcyclohexene but not cycloocta-1,5-diene.
Short Answer
Answer
Heating buta-1,3-diene forms 4-vinylcyclohexene but not cycloocta-1,5-diene as the formation of cycloocta-1,5-diene involves a cycloaddition reaction that is not thermally feasible.
The formation of 4-vinylcyclohexene involves the thermally feasible cycloaddition reaction.