Chapter 13: Q 5. (page 501)
Question: What molecular ions would you expect for the compound depicted in the ball-and-stick model?
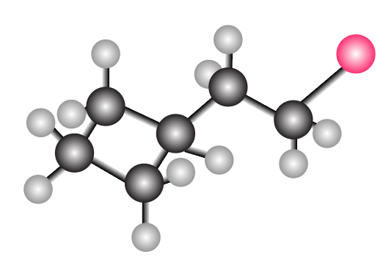
Short Answer
Answer
For the given compound, 162 and 164 molecular ions are expected.
Learning Materials
Features
Discover
Chapter 13: Q 5. (page 501)
Question: What molecular ions would you expect for the compound depicted in the ball-and-stick model?

Answer
For the given compound, 162 and 164 molecular ions are expected.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: How do the IR spectra of the isomers cyclopentane and pent-1-ene differ?
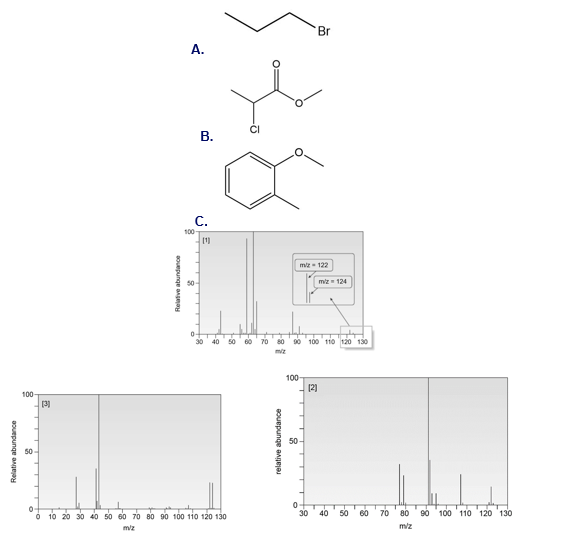
Question:Match each structure to its mass spectrum.
Question:Like alcohols, ethers undergo α cleavage by breaking a carbon–carbon bond between an alkyl group and the carbon bonded to the ether oxygen atom; that is, the red C–C bond in is broken. With this in mind, propose structures for the fragments formed by α cleavage of. Suggest a reason why an ether fragments by α cleavage.
Question:Propose four possible structures for a hydrocarbon with a molecular ion at m/z= 112.
Question:Propose a structure consistent with each set of data.
a. a compound that contains a benzene ring and has a molecular ion at m/z= 107
b. a hydrocarbon that contains only sp3 hybridized carbons and a molecular ion at m/z= 84
c. a compound that contains a carbonyl group and gives a molecular ion at m/z= 114
d. a compound that contains C, H, N, and O and has an exact mass for the molecular ion at
101.0841
What do you think about this solution?
We value your feedback to improve our textbook solutions.