Chapter 13: Q 50. (page 525)
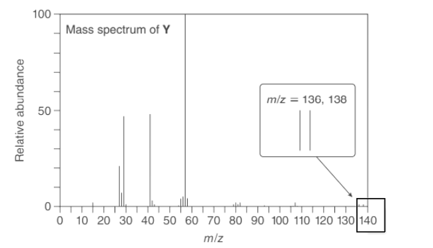
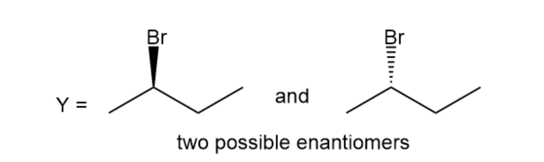
Question:A chiral compound Y has a strong absorption at 2970–2840 in its IR spectrum and gives the following mass spectrum. Propose a structure for Y.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 13: Q 50. (page 525)
Question:A chiral compound Y has a strong absorption at 2970–2840 in its IR spectrum and gives the following mass spectrum. Propose a structure for Y.

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question:Match each structure to its mass spectrum.

Question: What are the major IR absorptions in the functional group region for oleic acid, a common unsaturated fatty acid (Section 10.6A)?

Question: What is the mass of the molecular ion formed from compounds having each molecular formula:
(a) ; (b);(c); (d) methamphetamine ?
Question: What functional groups are responsible for the absorptions above 1500 in the IR spectra for compounds A and B?
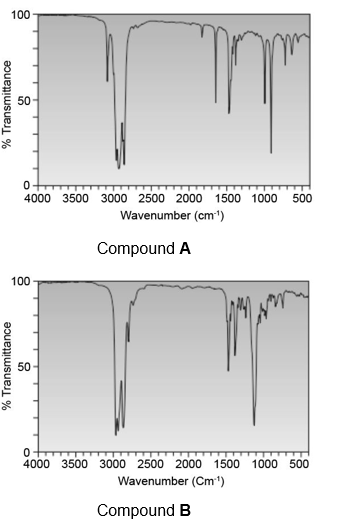
Question: Reaction of tert-butyl pentyl ether with HBr forms 1-bromopentane and compound B. B has a molecular ion in its mass spectrum at 56 and gives peaks in its IR spectrum at 3150–3000, 3000–2850, and 1650 cm-1. Propose a structure for B and draw a stepwise mechanism that accounts for its formation.
What do you think about this solution?
We value your feedback to improve our textbook solutions.