Chapter 13: Q 31. (page 521)
Question:Match each structure to its mass spectrum.
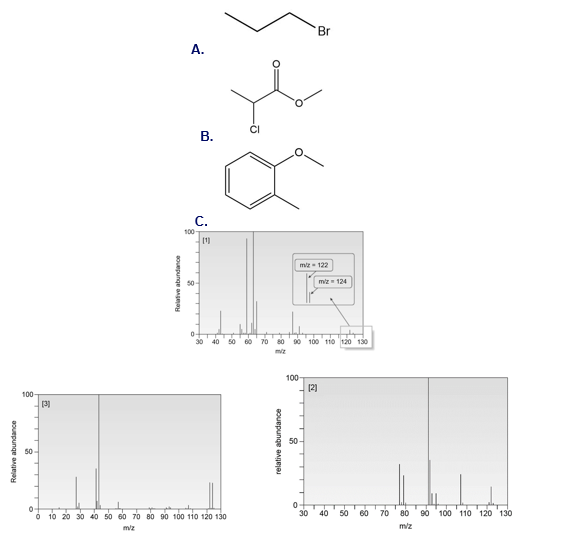
Short Answer
Answer
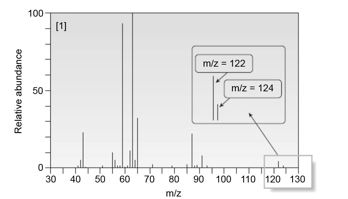
Compound A :
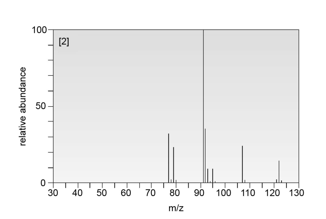
Compound B :
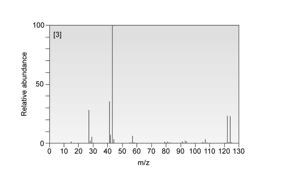
Compound C :
Learning Materials
Features
Discover
Chapter 13: Q 31. (page 521)
Question:Match each structure to its mass spectrum.

Answer
Compound A :

Compound B :

Compound C :

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: A chiral hydrocarbon X exhibits a molecular ion at 82 in its massspectrum. The IR spectrum of X shows peaks at 3300, 3000–2850, and 2250 cm-1. Propose a structure for X.
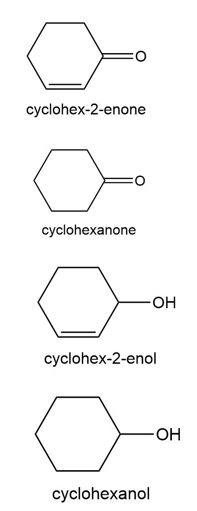
Question:Reduction of cyclohex-2-enone can yield cyclohexanone, cyclohex-2-enol, or cyclohexanol, depending on the reagent and reaction conditions. How could you use IR spectroscopy to distinguish the three possible products?
Question: Propose two molecular formulas for each molecular ion: (a) 102; (b) 98; (c) 119; (d) 74.
Question:A low-resolution mass spectrum of the neurotransmitter dopamine gave a molecular ion at m/z= 153. Two possible molecular formulas for this molecular ion are and . A high-resolution mass spectrum provided an exact mass at 153.0680. Which of the possible molecular formulas is the correct one?
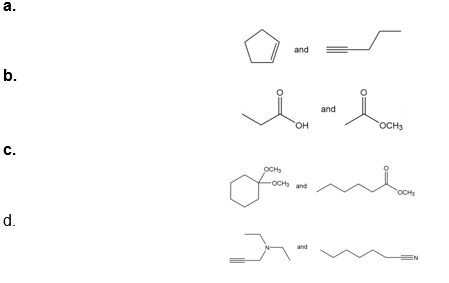
Question: How would each of the following pairs of compounds differ in their IR spectra?
What do you think about this solution?
We value your feedback to improve our textbook solutions.