Chapter 13: Q 49. (page 524)
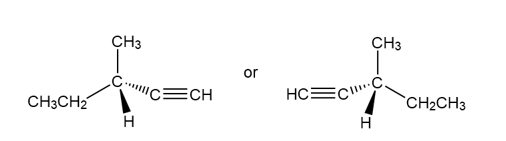
Question: A chiral hydrocarbon X exhibits a molecular ion at 82 in its massspectrum. The IR spectrum of X shows peaks at 3300, 3000–2850, and 2250 cm-1. Propose a structure for X.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 13: Q 49. (page 524)
Question: A chiral hydrocarbon X exhibits a molecular ion at 82 in its massspectrum. The IR spectrum of X shows peaks at 3300, 3000–2850, and 2250 cm-1. Propose a structure for X.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What major IR absorptions are present above 1500 for each compound?
Question: What is the mass of the molecular ion formed from compounds having each molecular formula:
(a) ; (b);(c); (d) methamphetamine ?
Question: Propose two molecular formulas for each of the following molecular ions: (a) 72; (b) 100; (c) 73.
Question: What molecular ions would you expect for compounds having each of the following molecular formulas: (a); (b); (c); (d)?
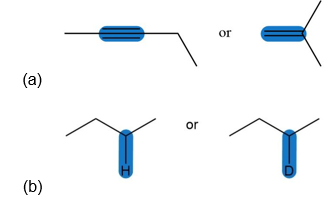
Question: Which highlighted bond in each pair absorbs at a higher wavenumber?
What do you think about this solution?
We value your feedback to improve our textbook solutions.