Chapter 13: Q 32. (page 521)
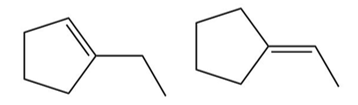
Question:Propose two possible structures for a hydrocarbon having an exact mass of 96.0939 that forms ethylcyclopentane upon hydrogenation with H2 and Pd-C.
Short Answer
Answer
Two possible structures:
Learning Materials
Features
Discover
Chapter 13: Q 32. (page 521)
Question:Propose two possible structures for a hydrocarbon having an exact mass of 96.0939 that forms ethylcyclopentane upon hydrogenation with H2 and Pd-C.
Answer
Two possible structures:

All the tools & learning materials you need for study success - in one app.
Get started for free
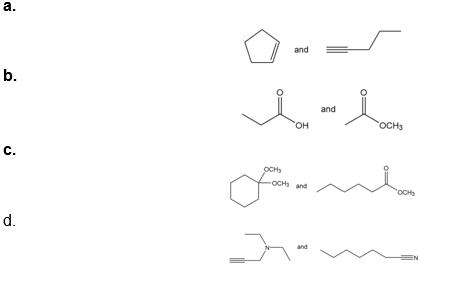
Question: How would each of the following pairs of compounds differ in their IR spectra?
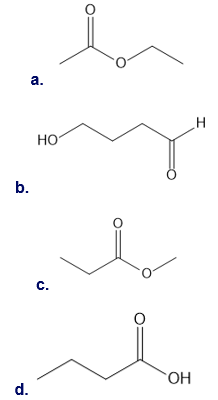
Question: Which of the following possible structures for X can be excluded on the basis of its IR spectrum?
Question:A low-resolution mass spectrum of the neurotransmitter dopamine gave a molecular ion at m/z= 153. Two possible molecular formulas for this molecular ion are and . A high-resolution mass spectrum provided an exact mass at 153.0680. Which of the possible molecular formulas is the correct one?
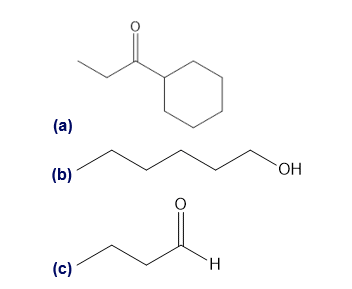
Question: What cations are formed in the mass spectrometer by α cleavage of each of the following compounds?
Question: Propose two molecular formulas for each of the following molecular ions: (a) 72; (b) 100; (c) 73.
What do you think about this solution?
We value your feedback to improve our textbook solutions.