Chapter 13: Q 9. (page 504)
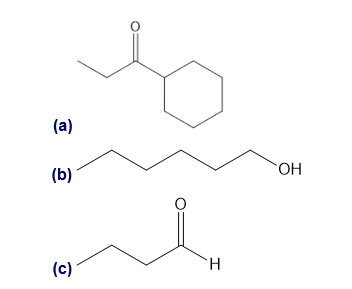
Question: What cations are formed in the mass spectrometer by α cleavage of each of the following compounds?
Short Answer
Answer

Learning Materials
Features
Discover
Chapter 13: Q 9. (page 504)
Question: What cations are formed in the mass spectrometer by α cleavage of each of the following compounds?

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
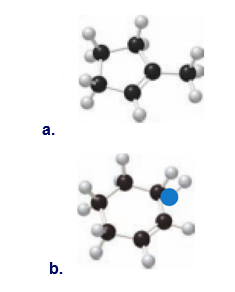
Question: What major IR absorptions are present above 1500 for each compound?
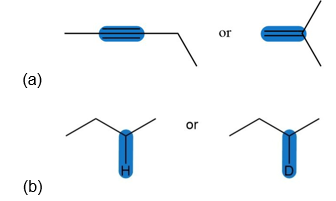
Question: Which highlighted bond in each pair absorbs at a higher wavenumber?
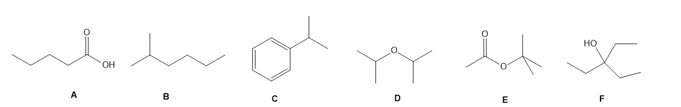
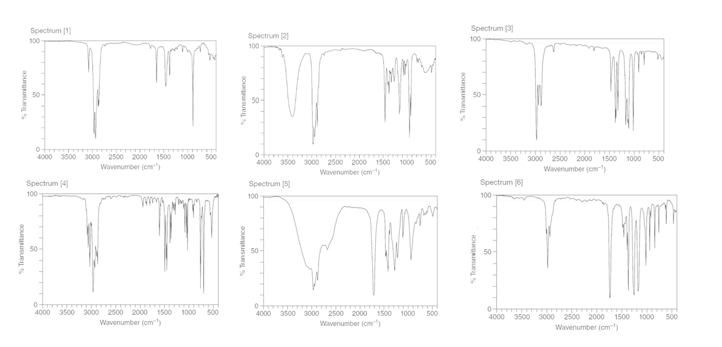
Question: Match each compound to its IR spectrum.
Question:Reaction of pentanoyl chloride with lithium dimethyl cuprate forms a compound J that has a molecular ion in its mass spectrum at 100, as well as fragments at m/z= 85, 57, and 43 (base). The IR spectrum of J has strong peaks at 2962 and 1718 cm–1. Propose a structure for J.
Question: The low-resolution mass spectrum of an unknown analgesic X had a molecular ion of 151. Possible molecular formulas include , , and . High-resolution mass spectrometry gave an exact mass of 151.0640. What is the molecular formula of X?
What do you think about this solution?
We value your feedback to improve our textbook solutions.