Chapter 13: Q 8. (page 504)
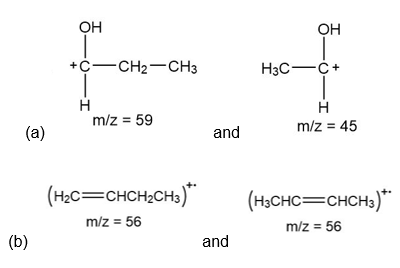
Question:(a) What mass spectral fragments are formed by α cleavage of butan-2-ol, ?
(b) What fragments are formed by dehydration of butan-2-ol?
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 13: Q 8. (page 504)
Question:(a) What mass spectral fragments are formed by α cleavage of butan-2-ol, ?
(b) What fragments are formed by dehydration of butan-2-ol?
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question:Benzonitrile (C6H5CN) is reduced to two different products depending on the reducing agent used. Treatment with lithium aluminum hydride followed by water forms K, which has a molecular ion in its mass spectrum at 107 and the following IR absorptions: 3373, 3290, 3062, 2920, and 1600 cm-1. Treatment with a milder reducing agent forms L, which has a molecular ion in its mass spectrum at 106 and the following IR absorptions: 3086, 2820, 2736, 1703, and 1600 cm-1. L shows fragments in its mass spectrum at m/z= 105 and 77. Propose structures for K and L and explain how you arrived at your conclusions.
Question: Which of the following has the higher frequency:(a) light having a wavelength of 102or 104 nm;
(b) light having a wavelength of 100 nm or 100 µm;
(c) red light or blue light?
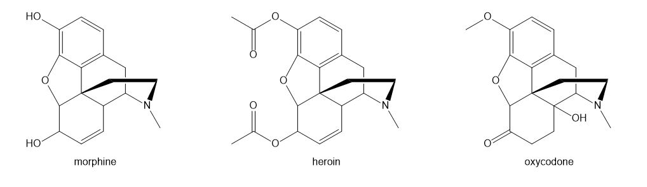
Question: Morphine, heroin, and oxycodone are three addicting analgesic narcotics. How could IR spectroscopy be used to distinguish these three compounds from each other?
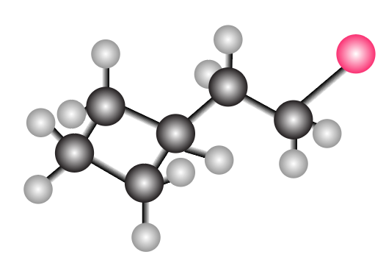
Question: What molecular ions would you expect for the compound depicted in the ball-and-stick model?
Question:For each compound, assign likely structures to the fragments at each m/zvalue, and explain how each fragment is formed.
a. : peaks at m/z= 104, 91
b. : peaks at m/z= 71, 68, 41, 31
What do you think about this solution?
We value your feedback to improve our textbook solutions.