Chapter 13: Q 33. (page 522)
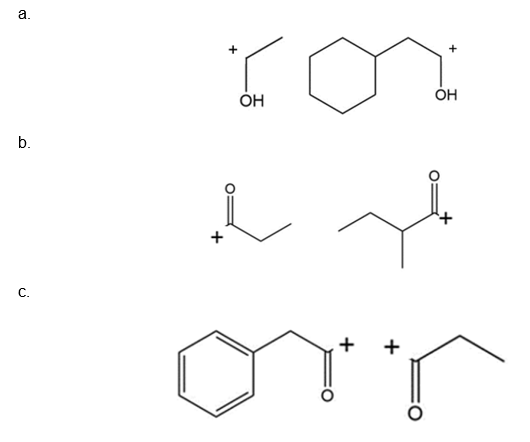
Question:What cations are formed in the mass spectrometer by α cleavage of each of the following compounds?
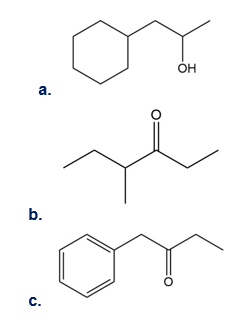
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 13: Q 33. (page 522)
Question:What cations are formed in the mass spectrometer by α cleavage of each of the following compounds?

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Treatment of benzaldehyde () with Zn(Hg) in aqueous HCl forms a compound Z that has a molecular ion at 92 in its mass spectrum. Z shows absorptions at 3150–2950, 1605, and 1496 cm-1 in its IR spectrum. Give a possible structure for Z.
Question: Propose structures consistent with each set of data: (a) a hydrocarbon with a molecular ion at m/z = 68 and IR absorptions at 3310, 3000–2850, and 2120 ;
(b) a compound containing C, H, and O with a molecular ion at m/z = 60 and IR absorptions at 3600–3200 and 3000–2850 .
Question:Propose a molecular formula for rose oxide, a rose-scented compound isolated from roses and geraniums, which contains the elements of C, H, and O, has two degrees of unsaturation, and a molecular ion in its mass spectrum at m/z= 154.
Question: Which of the highlighted bonds absorbs at higher in an IR spectrum?
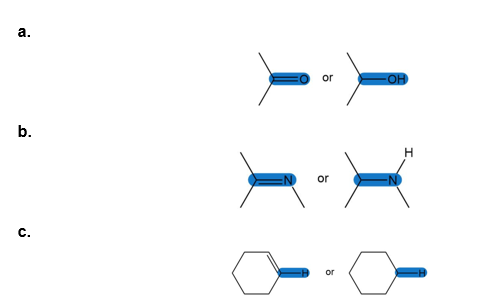
Question:Which compound gives a molecular ion at m/z= 122: , , or ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.