Chapter 13: Q 26. (page 521)
Question:Which compound gives a molecular ion at m/z= 122: , , or ?
Short Answer
Answer
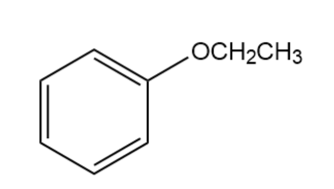
, Molecular ion peak atm/z = 122
Learning Materials
Features
Discover
Chapter 13: Q 26. (page 521)
Question:Which compound gives a molecular ion at m/z= 122: , , or ?
Answer

, Molecular ion peak atm/z = 122
All the tools & learning materials you need for study success - in one app.
Get started for free
Oxidation of citronellol, a constituent of rose and geranium oils, with PCC in the presence of added NaOCOCH3 forms compound A. A has a molecular ion in its mass spectrum at 154 and a strong peak in its IR spectrum at 1730 cm-1. Without added NaOCOCH3, oxidation of citronellol with PCC yields isopulegone, which is then converted to B with aqueous base. B has a molecular ion at 152, and a peak in its IR spectrum at 1680 cm-1.
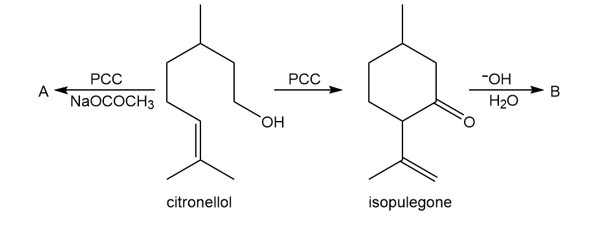
a. Identify the structures of A and B.
b. Draw a mechanism for the conversion of citronellol to isopulegone.
c. Draw a mechanism for the conversion of isopulegone to B.
Question:Like alcohols, ethers undergo α cleavage by breaking a carbon–carbon bond between an alkyl group and the carbon bonded to the ether oxygen atom; that is, the red C–C bond in is broken. With this in mind, propose structures for the fragments formed by α cleavage of. Suggest a reason why an ether fragments by α cleavage.
Question:Suppose you have two bottles, labeled ketone A and ketone B. You know that one bottle contains and one contains but you do not know which ketone is in which bottle. Ketone A gives a fragment at m/z= 99 and ketone B givesa fragment at m/z= 113. What are the likely structures of ketones A and B from these fragmentation data?
Question: Which of the following has higher energy:
(a) IR light of 3000 or 1500 in wavenumber;
(b) IR light having a wavelength of 10 µm or 20 µm?
Question:For each compound, assign likely structures to the fragments at each m/zvalue, and explain how each fragment is formed.
a. : peaks at m/z= 104, 91
b. : peaks at m/z= 71, 68, 41, 31
What do you think about this solution?
We value your feedback to improve our textbook solutions.