Chapter 21: 16P (page 817)
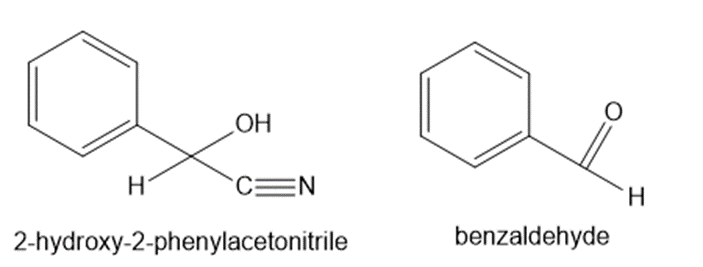
What cyanohydrin and carbonyl compound are formed when amygdalin is metabolized in a similar manner to linamarin?
Short Answer
The compounds are mentioned below:
Learning Materials
Features
Discover
Chapter 21: 16P (page 817)
What cyanohydrin and carbonyl compound are formed when amygdalin is metabolized in a similar manner to linamarin?
The compounds are mentioned below:

All the tools & learning materials you need for study success - in one app.
Get started for free
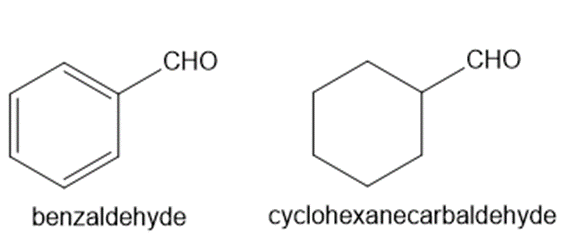
Explain why benzaldehyde is less reactive than cyclohexanecarbaldehyde towards nucleophilic attack.
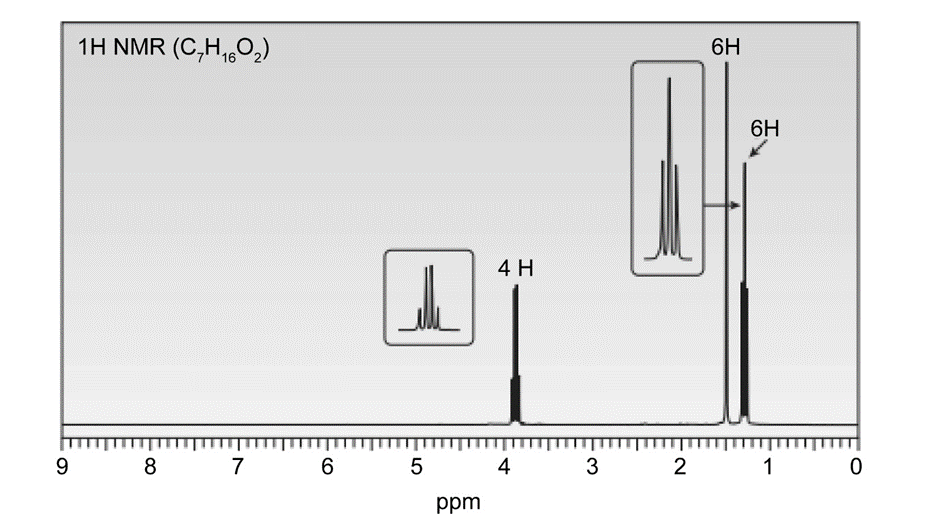
A solution of acetone (CH3)2C=O in ethanol CH3CH2OH in the presence of a trace of acid was allowed to stand for several days, and a new compound of molecular formula C7H16O2 was formed. The IR spectrum showed only one major peak in the functional group region around 3000 cm-1 , and the 1H-NMR spectrum is given here. What is the structure of the product?
Draw all stereoisomers formed in each reaction.
a.
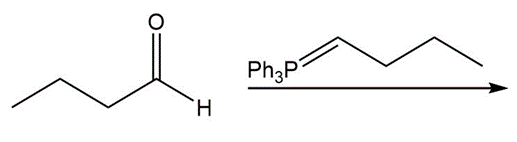
b.
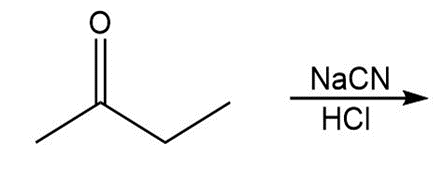
c.
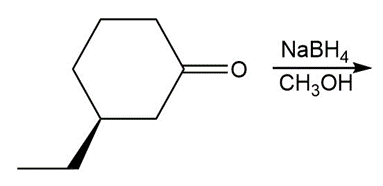
d.
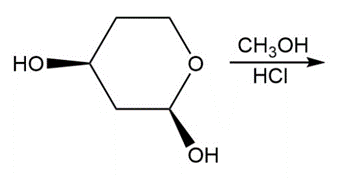
Draw a stepwise mechanism for the following reaction, a key step in the synthesis of conivaptan (trade name Vaprisol), a drug used in the treatment of low sodium levels.
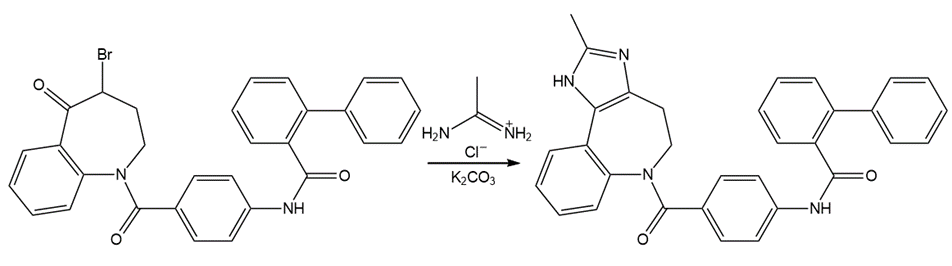
Draw a stepwise mechanism for the following reaction, a key step in the synthesis of the anti-inflammatory drug celecoxib (trade name Celebrex).

What do you think about this solution?
We value your feedback to improve our textbook solutions.