Chapter 21: 48P (page 817)
Draw all stereoisomers formed in each reaction.
a.
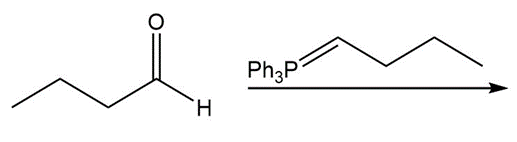
b.
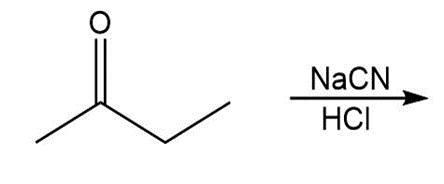
c.
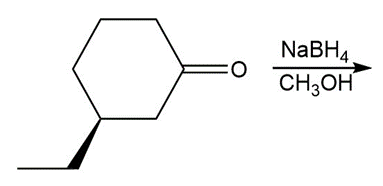
d.

Short Answer
a.
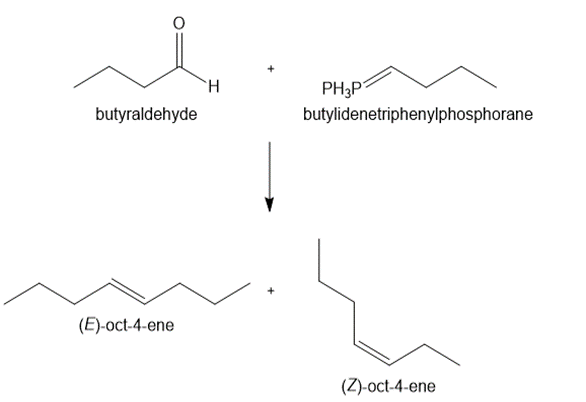
b.
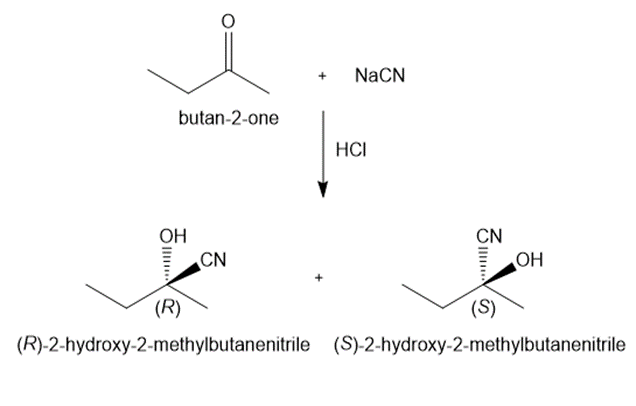
c.
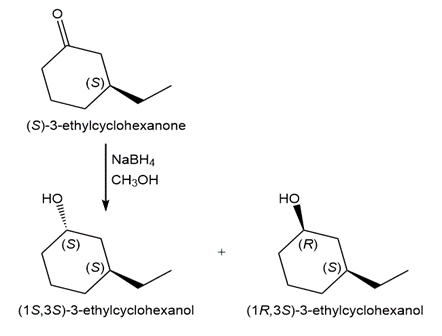
d.
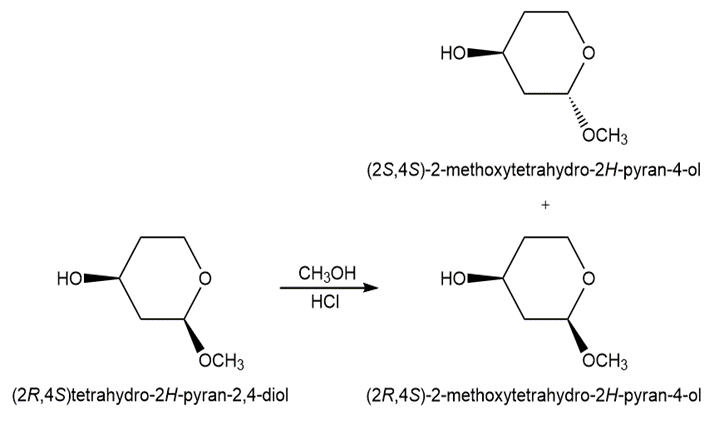
Learning Materials
Features
Discover
Chapter 21: 48P (page 817)
Draw all stereoisomers formed in each reaction.
a.
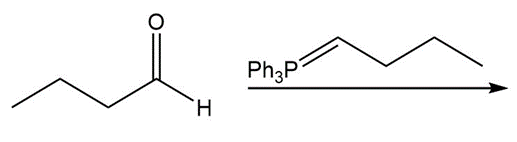
b.
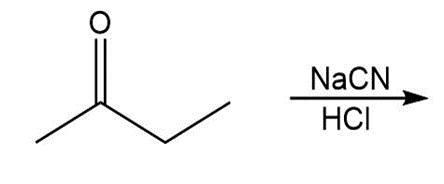
c.

d.

a.

b.

c.

d.

All the tools & learning materials you need for study success - in one app.
Get started for free
Devise a synthesis of each alkene using a Wittig reaction to form the double bond. You may use benzene and organic alcohols having four or fewer carbons as starting materials and any required reagents
a.
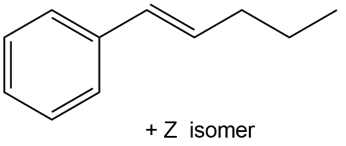
b.
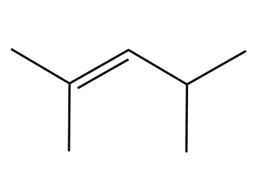
Draw a stepwise mechanism for the following imine hydrolysis
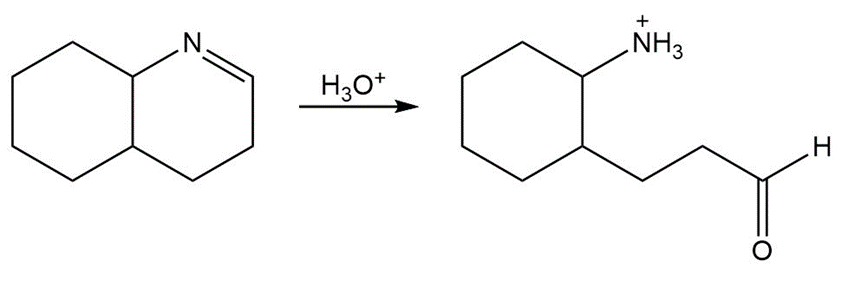
Rank the following compounds in order of increasing reactivity in nucleophilic addition
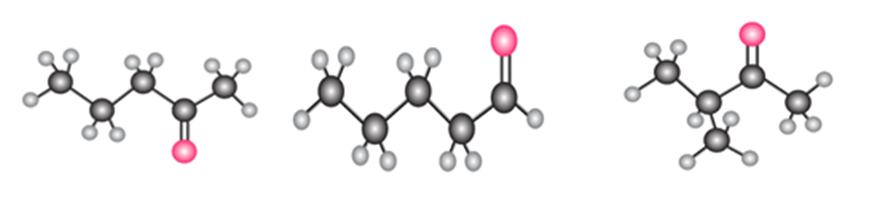
What products are formed when each acetal is hydrolyzed with aqueous acid?
a.
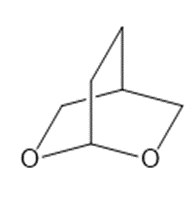
b.
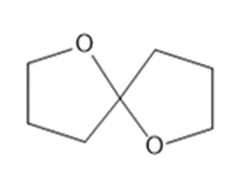
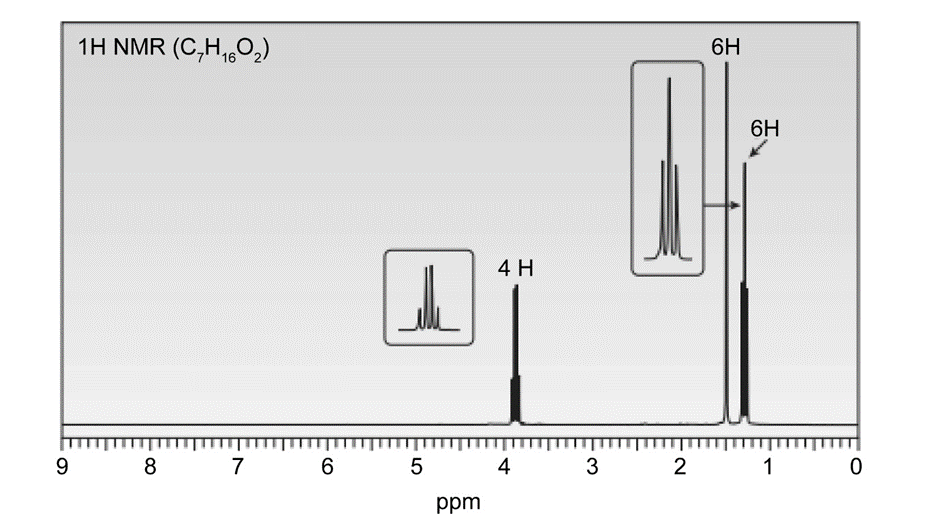
A solution of acetone (CH3)2C=O in ethanol CH3CH2OH in the presence of a trace of acid was allowed to stand for several days, and a new compound of molecular formula C7H16O2 was formed. The IR spectrum showed only one major peak in the functional group region around 3000 cm-1 , and the 1H-NMR spectrum is given here. What is the structure of the product?
What do you think about this solution?
We value your feedback to improve our textbook solutions.