Chapter 21: 41P (page 817)
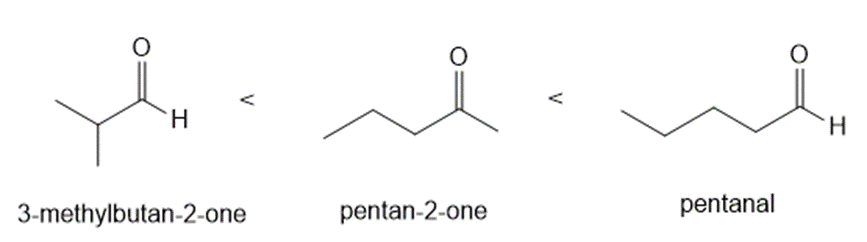
Rank the following compounds in order of increasing reactivity in nucleophilic addition

Short Answer
Learning Materials
Features
Discover
Chapter 21: 41P (page 817)
Rank the following compounds in order of increasing reactivity in nucleophilic addition


All the tools & learning materials you need for study success - in one app.
Get started for free
Reaction of 5,5-dimethoxypentan-2-one with methylmagnesium iodide followed by treatment with aqueous acid forms cyclic hemiacetal Y. Draw a stepwise mechanism that illustrates how Y is formed
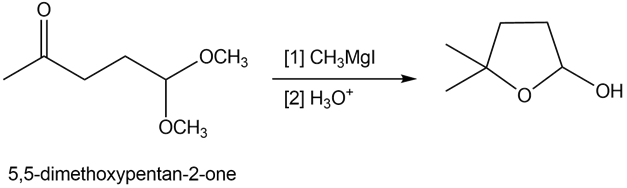
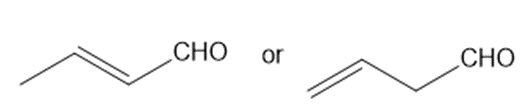
Which carbonyl group in each pair absorbs at a higher frequency?
a.
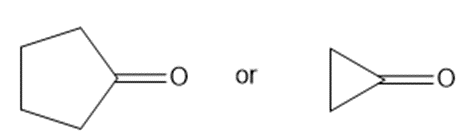
b.
Draw a stepwise mechanism for the following reaction, a key step in the synthesis of the anti-inflammatory drug celecoxib (trade name Celebrex).

The boiling point of butan-2-one ( 79.64oC ) is significantly higher than the boiling point of diethyl ether ( 34.6OC), even though both compounds exhibit dipole–dipole interactions and have comparable molecular weights. Offer an explanation.
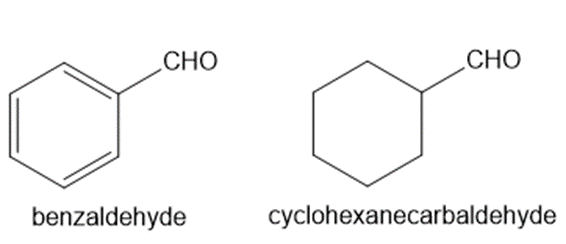
Explain why benzaldehyde is less reactive than cyclohexanecarbaldehyde towards nucleophilic attack.
What do you think about this solution?
We value your feedback to improve our textbook solutions.