Chapter 10: Q64 E (page 590)
What types of liquids typically form amorphous solids?
Short Answer
Large and cumbersome molecules are amorphous solid
Learning Materials
Features
Discover
Chapter 10: Q64 E (page 590)
What types of liquids typically form amorphous solids?
Large and cumbersome molecules are amorphous solid
All the tools & learning materials you need for study success - in one app.
Get started for free
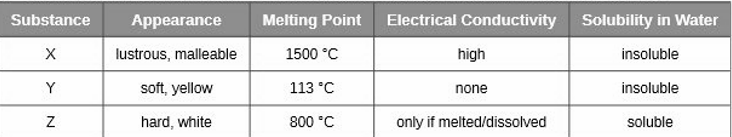
Classify each substance in the table as either a metallic, ionic, molecular, or covalent network solid
At very low temperatures oxygen, O2, freezes and forms a crystalline solid. Which best describes these crystals?
(a) ionic (b) covalent network (c) metallic (d) amorphous (e) molecular crystals
Question: In terms of their bulk properties, how do liquids and solids differ? How are they similar?
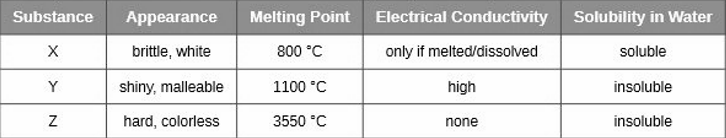
Classify each substance in the table as either a metallic, ionic, molecular, or covalent network solid
Carbon tetrachloride, \({\rm{CC}}{{\rm{l}}_{\rm{4}}}\), was once used as a dry-cleaning solvent, but is no longer used because it is carcinogenic. At \({\bf{57}}.{\bf{8}}\)°C, the vapor pressure of \({\rm{CC}}{{\rm{l}}_{\rm{4}}}\) is \({\bf{54}}.{\bf{0}}\)kPa, and its enthalpy of vaporization is \({\bf{33}}.{\bf{05}}\)kJ/mol. Use this information to estimate the normal boiling point for \({\rm{CC}}{{\rm{l}}_{\rm{4}}}\).
What do you think about this solution?
We value your feedback to improve our textbook solutions.