Chapter 10: Q70E. (page 591)
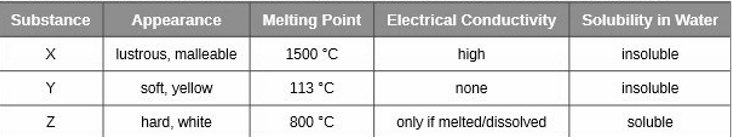
Classify each substance in the table as either a metallic, ionic, molecular, or covalent network solid
Short Answer
X is metallic, Y is covalent and Z is ionic solids.
Learning Materials
Features
Discover
Chapter 10: Q70E. (page 591)
Classify each substance in the table as either a metallic, ionic, molecular, or covalent network solid

X is metallic, Y is covalent and Z is ionic solids.
All the tools & learning materials you need for study success - in one app.
Get started for free
Platinum (atomic radius \( = 1.38{A^o}\) ) crystallizes in a cubic closely packed structure. Calculate the edge length of the face-centered cubic unit cell and the density of platinum.
What is the coordination number of a chromium atom in the body-centered cubic structure of chromium?
Question: In terms of the kinetic molecular theory, in what ways are liquids similar to gases? In what ways are liquids different from gases?
Which contains the compounds listed correctly in order of increasing boiling points
\(\begin{aligned}{\rm{(a)}}{{\rm{N}}_{\rm{2}}}{\rm{ < C}}{{\rm{S}}_{\rm{2}}}{\rm{ < }}{{\rm{H}}_{\rm{2}}}{\rm{O < KCl}}\\{\rm{(b)}}{{\rm{H}}_{\rm{2}}}{\rm{O < }}{{\rm{N}}_{\rm{2}}}{\rm{ < C}}{{\rm{S}}_{\rm{2}}}{\rm{ < KCl}}\\{\rm{(c)}}{{\rm{N}}_{\rm{2}}}{\rm{ < KCl < C}}{{\rm{S}}_{\rm{2}}}{\rm{ < }}{{\rm{H}}_{\rm{2}}}{\rm{O}}\\{\rm{(d)C}}{{\rm{S}}_{\rm{2}}}{\rm{ < }}{{\rm{N}}_{\rm{2}}}{\rm{ < KCl < }}{{\rm{H}}_{\rm{2}}}{\rm{O}}\\{\rm{(e)KCl < }}{{\rm{H}}_{\rm{2}}}{\rm{O < C}}{{\rm{S}}_{\rm{2}}}{\rm{ < }}{{\rm{N}}_{\rm{2}}}\end{aligned}\)
You may have heard someone use the figure of speech “slower than molasses in winter” to describe a process that occurs slowly. Explain why this is an apt idiom, using concepts of molecular size and shape, molecular interactions, and the effect of changing temperature.
What do you think about this solution?
We value your feedback to improve our textbook solutions.