Chapter 10: Q71 E (page 592)
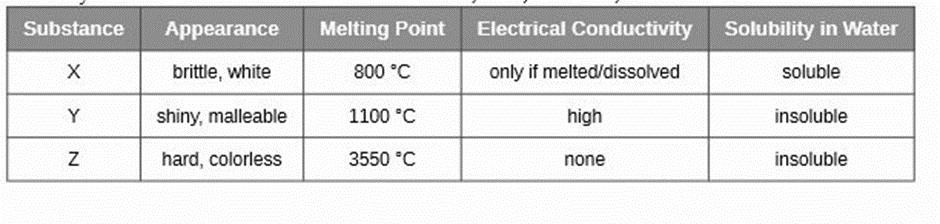
Classify each substance in the table as either a metallic, ionic, molecular, or covalent network solid
Short Answer
X is ionic, Y is metallic and Z is covalent network solids.
Learning Materials
Features
Discover
Chapter 10: Q71 E (page 592)
Classify each substance in the table as either a metallic, ionic, molecular, or covalent network solid

X is ionic, Y is metallic and Z is covalent network solids.
All the tools & learning materials you need for study success - in one app.
Get started for free
Heat is added to ice at 0 °C. Explain why the temperature of the ice does not change. What does change?
Identify the following substances as ionic, metallic, covalent network, or molecular solids: Substance A is malleable, ductile, conducts electricity well, and has a melting point of\({\rm{1135 ^\circ C}}\). Substance B is brittle, does not conduct electricity as a solid but does when molten, and has a melting point of\({\rm{2072 ^\circ C}}\). Substance C is very hard, does not conduct electricity, and has a melting point of\({\rm{3440 ^\circ C}}\). Substance D is soft, does not conduct electricity, and has a melting point of\({\rm{185 ^\circ C}}\).
How does the boiling of a liquid differ from its evaporation?
The molecular mass of butanol,\({C_4}{H_9}OH\), is\(74.14\); that of ethylene glycol, \({\rm{C}}{{\rm{H}}_{\rm{2}}}\left( {{\rm{OH}}} \right){\rm{C}}{{\rm{H}}_{\rm{2}}}{\rm{OH}}\), is 62.08, yet their boiling points are \({\bf{117}}.{\bf{2}}{\rm{ }}^\circ {\bf{C}}\) and \(174 ^\circ C,\)respectively. Explain the reason for the difference.
Dry ice, \({\rm{C}}{{\rm{O}}_{\rm{2}}}{\rm{(s)}}\), does not melt at atmospheric pressure. It sublimes at a temperature of \({\rm{ - 7}}{{\rm{8}}^{\rm{o}}}{\rm{C}}\). What is the lowest pressure at which \({\rm{C}}{{\rm{O}}_{\rm{2}}}{\rm{(s)}}\) will melt to give \({\rm{C}}{{\rm{O}}_{\rm{2}}}{\rm{(l)}}\)? At approximately what temperature will this occur? (See Figure \({\rm{10}}{\rm{.34}}\) for the phase diagram.)
What do you think about this solution?
We value your feedback to improve our textbook solutions.