Chapter 10: Q71E. (page 592)
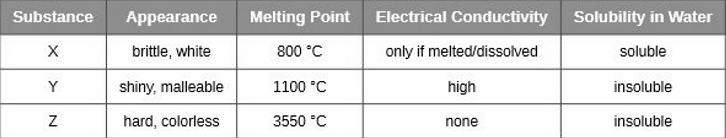
Classify each substance in the table as either a metallic, ionic, molecular, or covalent network solid
Short Answer
X is ionic, Y is metallic and Z is covalent network solids.
Learning Materials
Features
Discover
Chapter 10: Q71E. (page 592)
Classify each substance in the table as either a metallic, ionic, molecular, or covalent network solid

X is ionic, Y is metallic and Z is covalent network solids.
All the tools & learning materials you need for study success - in one app.
Get started for free
The density of aluminium is \(2.7\;{\rm{g}}/{\rm{c}}{{\rm{m}}^3}\); that of silicon is \(2.3\;{\rm{g}}/{\rm{c}}{{\rm{m}}^3}\). Explain why Si has the lower density even though it has heavier atoms.
Determine the phase changes that carbon dioxide undergoes as the pressure changes if the temperature is held at −50°C? If the temperature is held at −40°C? At 20°C?
Identify the type of crystalline solid (metallic, network covalent, ionic, or molecular) formed by each of the following substances:
(a) SiO2
(b) KCl
(c) Cu
(d) CO2
(e) C(Diamond)
(f) BaSO4
(g) NH3
(h) NH4F
(i) C2H5OH
What types of liquids typically form amorphous solids?
Identify two common observations indicating some liquids have sufficient vapor pressures to noticeably evaporate.
What do you think about this solution?
We value your feedback to improve our textbook solutions.