Chapter 10: Q64E. (page 590)
What types of liquids typically form amorphous solids?
Short Answer
Large and cumbersome molecules are amorphous solid
Learning Materials
Features
Discover
Chapter 10: Q64E. (page 590)
What types of liquids typically form amorphous solids?
Large and cumbersome molecules are amorphous solid
All the tools & learning materials you need for study success - in one app.
Get started for free
Determine the phase changes that carbon dioxide undergoes as the pressure changes if the temperature is held at −50°C? If the temperature is held at −40°C? At 20°C?
Ammonia can be liquefied by compression at room temperature; oxygen cannot be liquefied under these conditions. Why do the two gases exhibit different behaviour?
Describe the crystal structure of Pt, which crystallizes with four equivalent metal atoms in a cubic unit cell
The surface tension and viscosity values for diethyl ether, acetone, ethanol, and ethylene glycol are shown here
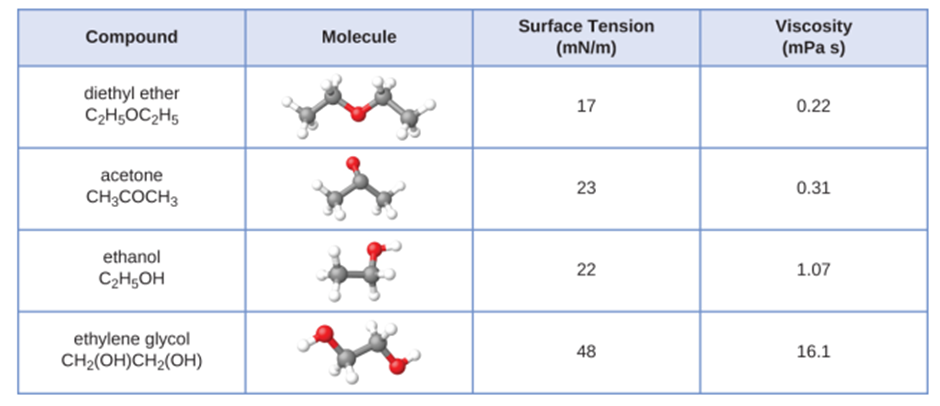
(a) Explain their differences in viscosity in terms of the size and shape of their molecules and their IMFs.
(b) Explain their differences in surface tension in terms of the size and shape of their molecules and their IMFs:
As it cools, olive oil slowly solidifies and forms a solid over a range of temperatures. Which best describes the solid?
(a) ionic (b) covalent network (c) metallic (d) amorphous (e) molecular crystals
What do you think about this solution?
We value your feedback to improve our textbook solutions.