Chapter 10: Q10.3-40 E (page 587)
How does the boiling of a liquid differ from its evaporation?
Short Answer
The boiling of a liquid differs from its evaporation, as evaporation is the term used to describe a relatively slow vaporization process.
Learning Materials
Features
Discover
Chapter 10: Q10.3-40 E (page 587)
How does the boiling of a liquid differ from its evaporation?
The boiling of a liquid differs from its evaporation, as evaporation is the term used to describe a relatively slow vaporization process.
All the tools & learning materials you need for study success - in one app.
Get started for free
At 250C, how high will water rise in a glass capillary tube with an inner diameter of 0.63 mm?
The free space in a metal may be found by subtracting the volume of the atoms in a unit cell from the volume of the cell. Calculate the percentage of free space in each of the three cubic lattices if all atoms in each are of equal size and touch their nearest neighbours. Which of these structures represents the most efficient packing? That is, which one of them packs with the least amount of unused space?
Ethyl chloride is used as a local anesthetic. When the liquid is sprayed on the skin, it cools the skin enough to freeze and numb it. Explain the cooling effect of liquid ethyl chloride.
Identify the following substances as ionic, metallic, covalent network, or molecular solids: Substance A is malleable, ductile, conducts electricity well, and has a melting point of\({\rm{1135 ^\circ C}}\). Substance B is brittle, does not conduct electricity as a solid but does when molten, and has a melting point of\({\rm{2072 ^\circ C}}\). Substance C is very hard, does not conduct electricity, and has a melting point of\({\rm{3440 ^\circ C}}\). Substance D is soft, does not conduct electricity, and has a melting point of\({\rm{185 ^\circ C}}\).
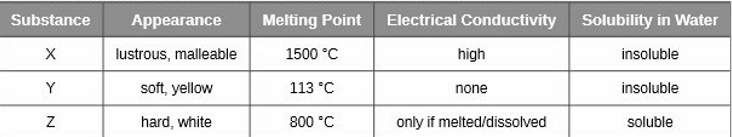
Classify each substance in the table as either a metallic, ionic, molecular, or covalent network solid
What do you think about this solution?
We value your feedback to improve our textbook solutions.