Chapter 10: Q10.3-41 E (page 587)
Use the information to estimate the boiling point of water in Denver when the atmospheric pressure is \(83.3\)kPa
Short Answer
The boiling point of water in Denver is\({\rm{9}}{{\rm{5}}^{\rm{^\circ }}}{\rm{C}}\).
Learning Materials
Features
Discover
Chapter 10: Q10.3-41 E (page 587)
Use the information to estimate the boiling point of water in Denver when the atmospheric pressure is \(83.3\)kPa
The boiling point of water in Denver is\({\rm{9}}{{\rm{5}}^{\rm{^\circ }}}{\rm{C}}\).
All the tools & learning materials you need for study success - in one app.
Get started for free
The melting point of H2O(s) is \({\rm{0}}\)°C. Would you expect the melting point of H2S(s) to be \({\rm{ - 8}}{{\rm{5}}^{\rm{^\circ }}}\)C, \({{\rm{0}}^{\rm{^\circ }}}\)C, or \({\rm{ - 18}}{{\rm{5}}^{\rm{o}}}\)C? Explain your answer.
Ammonia can be liquefied by compression at room temperature; oxygen cannot be liquefied under these conditions. Why do the two gases exhibit different behaviour?
Explain why the enthalpies of vaporization of the following substances increase in the order CH4 < NH3 < H2O, even though all three substances have approximately the same molar mass.
Question: In terms of the kinetic molecular theory, in what ways are liquids similar to gases? In what ways are liquids different from gases?
The surface tension and viscosity of water at several different temperatures are given in this table.
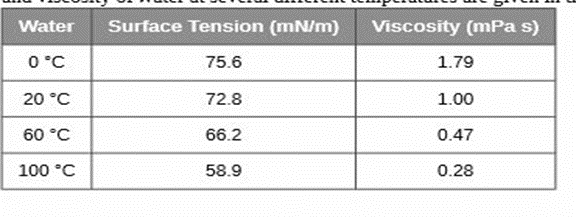
(a) As temperature increases, what happens to the surface tension of water? Explain why this occurs, in terms of molecular interactions and the effect of changing temperature.
(b) As temperature increases, what happens to the viscosity of water? Explain why this occurs, in terms of molecular interactions and the effect of changing temperature.
What do you think about this solution?
We value your feedback to improve our textbook solutions.