Chapter 10: Q70 E (page 591)
Classify each substance in the table as either a metallic, ionic, molecular, or covalent network solid
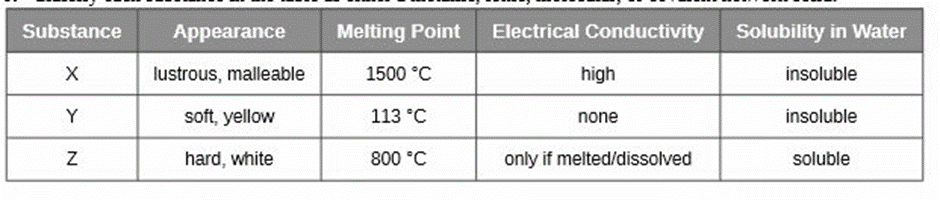
Short Answer
X is metallic, Y is covalent and Z is ionic solids.
Learning Materials
Features
Discover
Chapter 10: Q70 E (page 591)
Classify each substance in the table as either a metallic, ionic, molecular, or covalent network solid

X is metallic, Y is covalent and Z is ionic solids.
All the tools & learning materials you need for study success - in one app.
Get started for free
Ammonia can be liquefied by compression at room temperature; oxygen cannot be liquefied under these conditions. Why do the two gases exhibit different behaviour?
How much heat is required to evaporate 100.0 g of liquid ammonia, NH3, at its boiling point if its enthalpy of vaporization is 4.8 kJ/mol?
Explain why ice, which is a crystalline solid, has a melting temperature of 0 °C, whereas butter, which is an amorphous solid, softens over a range of temperatures.
How much heat is required to convert \({\rm{422 g}}\) of liquid \({{\rm{H}}_{\rm{2}}}{\rm{O}}\) at \({\rm{2}}{{\rm{3}}^{\rm{o}}}{\rm{C}}\) into steam at \({\rm{15}}{{\rm{0}}^{\rm{o}}}{\rm{C}}\)?
Heat is added to ice at 0 °C. Explain why the temperature of the ice does not change. What does change?
What do you think about this solution?
We value your feedback to improve our textbook solutions.