Chapter 10: Q1E (page 583)
Question: In terms of their bulk properties, how do liquids and solids differ? How are they similar?
Short Answer
Solids and liquids are similar in some cases but differ in their bulk properties.
Learning Materials
Features
Discover
Chapter 10: Q1E (page 583)
Question: In terms of their bulk properties, how do liquids and solids differ? How are they similar?
Solids and liquids are similar in some cases but differ in their bulk properties.
All the tools & learning materials you need for study success - in one app.
Get started for free
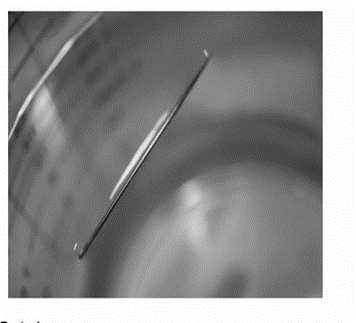
Although steel is denser than water, a steel needle or paper clip placed carefully lengthwise on the surface of still water can be made to float. Explain at a molecular level how this is possible:
As it cools, olive oil slowly solidifies and forms a solid over a range of temperatures. Which best describes the solid?
(a) ionic (b) covalent network (c) metallic (d) amorphous (e) molecular crystals
Water rises in a glass capillary tube to a height of 8.4cm. What is the diameter of the capillarytube?
Cobalt metal crystallizes in a hexagonal closest packed structure. What is the coordination number of a cobalt atom?
In terms of their bulk properties, how do liquids and solids differ? How are they similar?
What do you think about this solution?
We value your feedback to improve our textbook solutions.