Chapter 8: Q7 CYL (page 443)
How many unpaired electrons would be present on a Be22- ion? Would it be paramagnetic or diamagnetic?
Short Answer
It is paramagnetic and has two unpaired electrons.
Learning Materials
Features
Discover
Chapter 8: Q7 CYL (page 443)
How many unpaired electrons would be present on a Be22- ion? Would it be paramagnetic or diamagnetic?
It is paramagnetic and has two unpaired electrons.
All the tools & learning materials you need for study success - in one app.
Get started for free
For many years after they were discovered, it was believed that the noble gases could not form compounds. Now we know that belief to be incorrect. A mixture of xenon and fluorine gases, confined in a quartz bulb and placed on a windowsill, is found to slowly produce a white solid. Analysis of the compound indicates that it contains \({\rm{77}}{\rm{.55\% }}\)Xe and \({\rm{22}}{\rm{.45\% \;F}}\)by mass.
(a) What is the formula of the compound?
(b) Write a Lewis structure for the compound.
(c) Predict the shape of the molecules of the compound.
(d) What hybridization is consistent with the shape you predicted?
Strike-anywhere matches contain a layer of \({\rm{KCl}}{{\rm{O}}_{\rm{3}}}\) and a layer of \({{\rm{P}}_{\rm{4}}}{{\rm{S}}_{\rm{3}}}\). The heat produced by the friction of striking the match causes these two compounds to react vigorously, which sets fire to the wooden stem of the match. \({\rm{KCl}}{{\rm{O}}_{\rm{3}}}\) contains the \({\rm{Cl}}{{\rm{O}}_{\rm{3}}}^{\rm{ - }}\) ion. \({{\rm{P}}_{\rm{4}}}{{\rm{S}}_{\rm{3}}}\) is an unusual molecule with the skeletal structure.
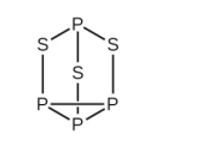
How many \({\rm{\sigma }}\)and \({\rm{\pi }}\)bonds are present in the molecule HCN?
Predict whether the MO diagram for S2 would show s-p mixing or not.
Explain why N22+ is diamagnetic, while O24+, which has the same number of valence electrons, is paramagnetic.
What do you think about this solution?
We value your feedback to improve our textbook solutions.