Chapter 8: Q19 E (page 448)
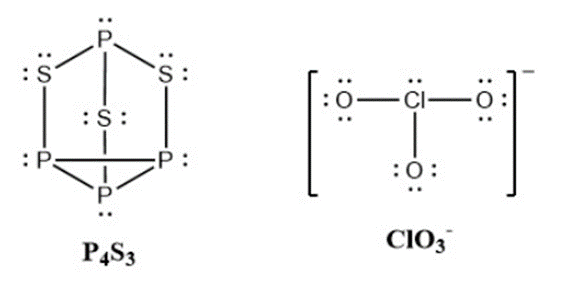
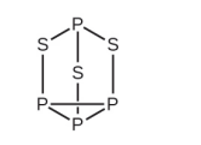
Strike-anywhere matches contain a layer of \({\rm{KCl}}{{\rm{O}}_{\rm{3}}}\) and a layer of \({{\rm{P}}_{\rm{4}}}{{\rm{S}}_{\rm{3}}}\). The heat produced by the friction of striking the match causes these two compounds to react vigorously, which sets fire to the wooden stem of the match. \({\rm{KCl}}{{\rm{O}}_{\rm{3}}}\) contains the \({\rm{Cl}}{{\rm{O}}_{\rm{3}}}^{\rm{ - }}\) ion. \({{\rm{P}}_{\rm{4}}}{{\rm{S}}_{\rm{3}}}\) is an unusual molecule with the skeletal structure.
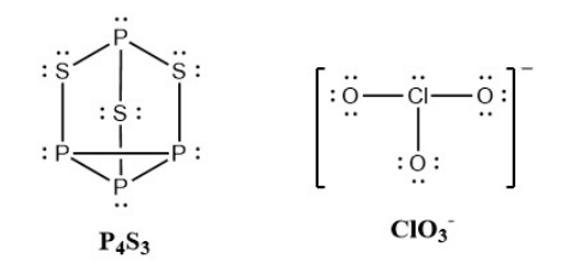
- Write Lewis structures for \({{\rm{P}}_{\rm{4}}}{{\rm{S}}_{\rm{3}}}\) and the \({\rm{Cl}}{{\rm{O}}_{\rm{3}}}^{\rm{ - }}\) ion.
- Describe the geometry about the \({\rm{P}}\) atoms, the \({\rm{S}}\) atom, and the \({\rm{Cl}}\) atom in these species.
- Assign a hybridization to the \({\rm{P}}\) atoms, the \({\rm{S}}\)atom, and the \({\rm{Cl}}\) atom in these species.
- Determine the oxidation states and formal charge of the atoms in \({{\rm{P}}_{\rm{4}}}{{\rm{S}}_{\rm{3}}}\) and the \({\rm{Cl}}{{\rm{O}}_{\rm{3}}}^{\rm{ - }}\) ion.
Short Answer
(a)
(b) P's geometry is trigonal pyramidal, S's geometry is bent, and Cl's geometry is trigonal pyramidal.
(c) P, S, and Cl have\({\rm{s}}{{\rm{p}}^{\rm{3}}}\)hybridization due to their tetrahedral electron-pair geometry.
(d) \({\rm{P}}\) has an oxidation state of \({\rm{ + 1}}\), \({\rm{S}}\) has \({\rm{ - }}\frac{{\rm{4}}}{{\rm{3}}}\), while \({\rm{Cl}}\) has \({\rm{ + 5}}\).\({\rm{P}}\) has a formal charge of \({\rm{0}}\), \({\rm{S}}\) has \({\rm{0}}\), \({\rm{Cl}}\) has \({\rm{ + 2}}\), and \({\rm{O}}\) has \({\rm{ - 1}}\)