Chapter 8: Q48 E (page 451)
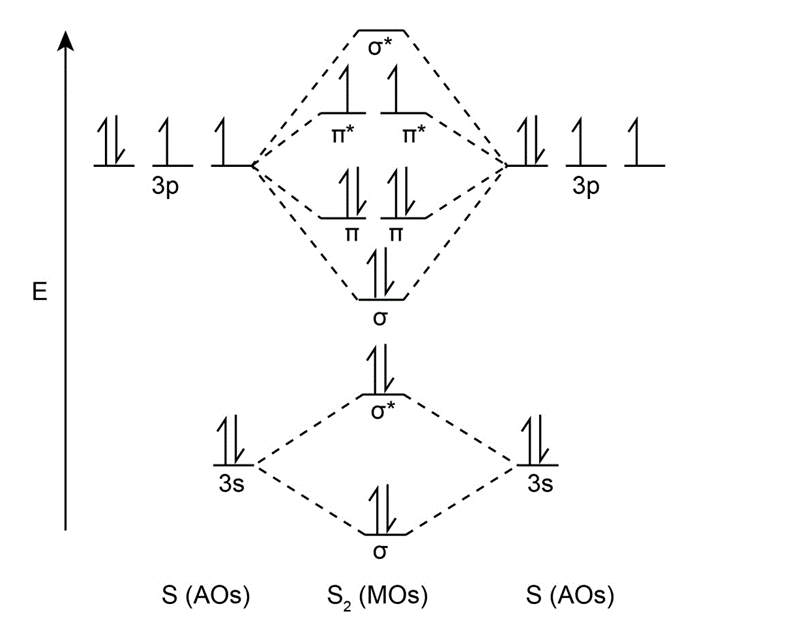
Predict whether the MO diagram for S2 would show s-p mixing or not.
Short Answer
The molecular orbital diagram for S2 would not show s-p mixing.
Learning Materials
Features
Discover
Chapter 8: Q48 E (page 451)
Predict whether the MO diagram for S2 would show s-p mixing or not.
The molecular orbital diagram for S2 would not show s-p mixing.

All the tools & learning materials you need for study success - in one app.
Get started for free
The bond energy of a C–C single bond averages \({\rm{347 kJ mo}}{{\rm{l}}^{{\rm{ - 1}}}}\); that of a
C ≡ C triple bond averages \({\rm{839 kJ mo}}{{\rm{l}}^{{\rm{ - 1}}}}\). Explain why the triple bond is not three times as strong as a single bond.
Using the MO diagrams, predict the bond order for the stronger bond in each pair:
B2or B2+
F2orF2+
O2 or O22+
C2+or C2-
Identify the hybridization of the central atom in each of the following molecules and ions that contain multiple bonds: (a) \({\rm{ClNO}}\) (\({\rm{N}}\) is the central atom) (b) \({\rm{C}}{{\rm{S}}_{\rm{2}}}\) (c) \({\rm{C}}{{\rm{l}}_{\rm{2}}}{\rm{CO}}\) (\({\rm{C}}\) is the central atom) (d) \({\rm{C}}{{\rm{l}}_{\rm{2}}}{\rm{SO}}\) (\({\rm{S}}\) is the central atom) (e) \({\rm{S}}{{\rm{O}}_{\rm{2}}}{{\rm{F}}_{\rm{2}}}\) (\({\rm{S}}\) is the central atom) (f) \({\rm{Xe}}{{\rm{O}}_{\rm{2}}}{{\rm{F}}_{\rm{2}}}\) (\({\rm{Xe}}\) is the central atom) (g) \({\rm{ClO}}{{\rm{F}}_{\rm{2}}}^{\rm{ + }}\) (\({\rm{Cl}}\) is the central atom).
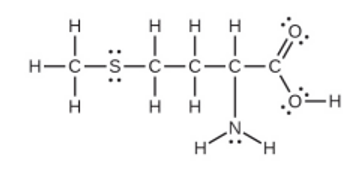
Methionine, \({\rm{C}}{{\rm{H}}_{\rm{3}}}{\rm{SC}}{{\rm{H}}_{\rm{2}}}{\rm{C}}{{\rm{H}}_{\rm{2}}}{\rm{CH}}\left( {{\rm{N}}{{\rm{H}}_{\rm{2}}}} \right){\rm{C}}{{\rm{O}}_{\rm{2}}}{\rm{H}}\), is an amino acid found in proteins. Draw a Lewis structure of this compound. What is the hybridization type of each carbon, oxygen, the nitrogen, and the sulfur?
Write Lewis structures for \({\rm{N}}{{\rm{F}}_{\rm{3}}}\) and \({\rm{P}}{{\rm{F}}_{\rm{5}}}\). On the basis of hybrid orbitals, explain the fact that \({\rm{N}}{{\rm{F}}_{\rm{3}}}\), \({\rm{P}}{{\rm{F}}_{\rm{3}}}\), and \({\rm{P}}{{\rm{F}}_{\rm{5}}}\) are stable molecules, but \({\rm{N}}{{\rm{F}}_{\rm{5}}}\) does not exist.
What do you think about this solution?
We value your feedback to improve our textbook solutions.