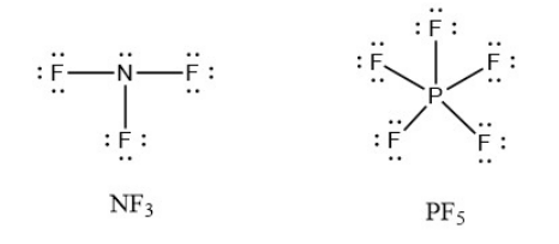Chapter 8: Q21 E (page 448)
Write Lewis structures for \({\rm{N}}{{\rm{F}}_{\rm{3}}}\) and \({\rm{P}}{{\rm{F}}_{\rm{5}}}\). On the basis of hybrid orbitals, explain the fact that \({\rm{N}}{{\rm{F}}_{\rm{3}}}\), \({\rm{P}}{{\rm{F}}_{\rm{3}}}\), and \({\rm{P}}{{\rm{F}}_{\rm{5}}}\) are stable molecules, but \({\rm{N}}{{\rm{F}}_{\rm{5}}}\) does not exist.
Short Answer
Nitrogen has four electron density zones in \({\rm{N}}{{\rm{F}}_{\rm{3}}}\) and phosphorus has four electron density regions in \({\rm{P}}{{\rm{F}}_{\rm{3}}}\) and is \({\rm{s}}{{\rm{p}}^{\rm{3}}}\) hybridised. Nitrogen and phosphorus will have five areas of electron density in \({\rm{N}}{{\rm{F}}_{\rm{5}}}\) and \({\rm{P}}{{\rm{F}}_{\rm{5}}}\), and will need to be \({\rm{s}}{{\rm{p}}^{\rm{3}}}{\rm{d}}\) hybridised, but nitrogen lacks a valence d orbital to accommodate the additional pair of electrons, whereas phosphorus does. So, whereas \({\rm{N}}{{\rm{F}}_{\rm{3}}}\), \({\rm{P}}{{\rm{F}}_{\rm{3}}}\), and \({\rm{P}}{{\rm{F}}_{\rm{5}}}\) are stable molecules, \({\rm{N}}{{\rm{F}}_{\rm{5}}}\) does not.