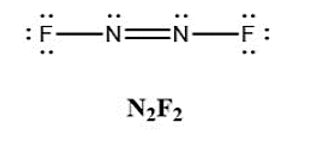Chapter 8: Q22 E (page 448)
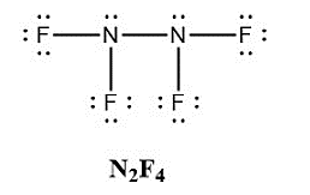
In addition to \({\rm{N}}{{\rm{F}}_{\rm{3}}}\), two other fluoro derivatives of nitrogen are known: \({{\rm{N}}_{\rm{2}}}{{\rm{F}}_{\rm{4}}}\) and \({{\rm{N}}_{\rm{2}}}{{\rm{F}}_{\rm{2}}}\). What shapes do you predict for these two molecules? What is the hybridization for the nitrogen in each molecule?
Short Answer
Nitrogens are\({\rm{s}}{{\rm{p}}^{\rm{3}}}\)hybridised in\({{\rm{N}}_{\rm{2}}}{{\rm{F}}_{\rm{4}}}\)and form a trigonal pyramidal structure.
Nitrogens in \({{\rm{N}}_{\rm{2}}}{{\rm{F}}_{\rm{2}}}\) are \({\rm{s}}{{\rm{p}}^{\rm{3}}}\) hybridised and have a bent form.