Chapter 8: Q5 E (page 446)
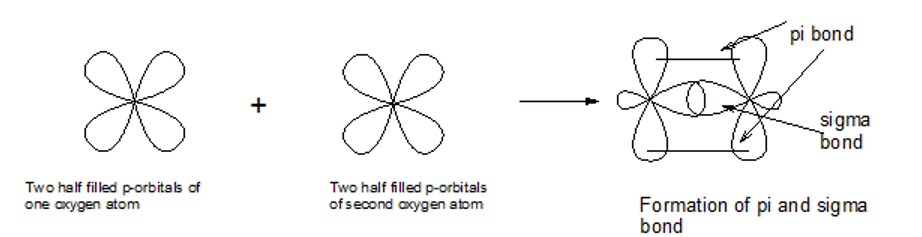
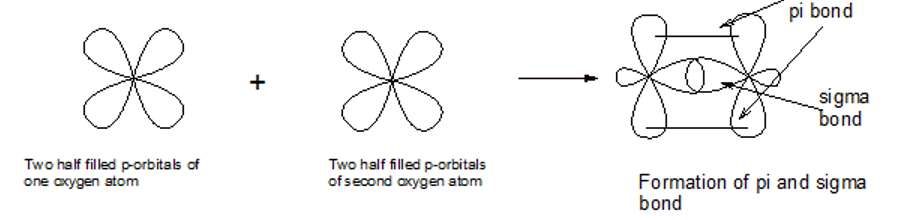
Use valence bond theory to explain the bonding in\({{\rm{O}}_{\rm{2}}}\). Sketch the overlap of the atomic orbitals involved in the bonds in \({{\rm{O}}_{\rm{2}}}\).
Short Answer
The molecules of oxygen contains a double bond consisting of sigma and pi bond.