Chapter 2: Q15CP (page 39)
What is the relationship between the strength of an acid and its pK value?
Short Answer
The and the strength of the acid is inversely proportional to each other.
Learning Materials
Features
Discover
Chapter 2: Q15CP (page 39)
What is the relationship between the strength of an acid and its pK value?
The and the strength of the acid is inversely proportional to each other.
All the tools & learning materials you need for study success - in one app.
Get started for free
You need a buffer at for use in purifying a protein at . You have chosen Tris, . You carefully make up Tris buffer, at data-custom-editor="chemistry" , and store it in the cold to equilibrate it to the temperature of the purification. When you measure the of the temperature-equilibrated buffer it has increased to . What is the explanation for this increase? How can you avoid this problem?
Question: Draw the structures of the conjugate bases of the following acids:
a.
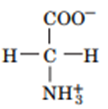
b.
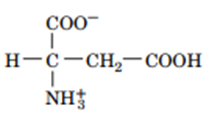
Describe the osmotic challenges facing a cell placed in pure water or in a high-salt solution.
(a) At any instant, how many water molecules are ionized in 1 L of pure water at pH 7.0? (b) Express this number as a percentage of the total water molecules.
Be able to sketch a titration curve and label its parts for a monoprotic and a polyprotic acid.
What do you think about this solution?
We value your feedback to improve our textbook solutions.