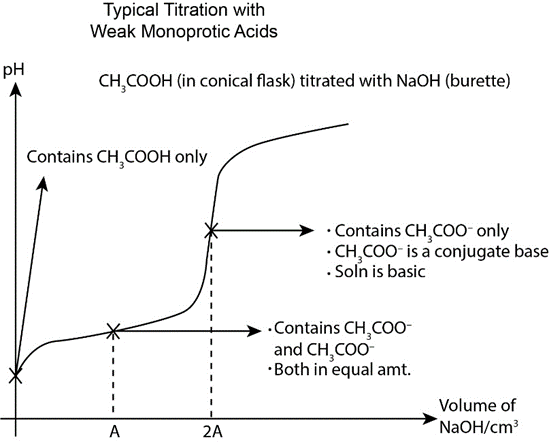
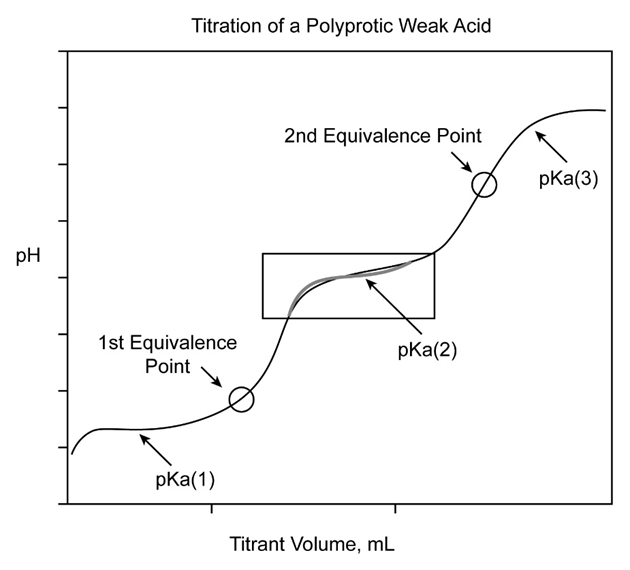
Chapter 2: Q17CP (page 39)
Be able to sketch a titration curve and label its parts for a monoprotic and a polyprotic acid.
Short Answer
Titrations are frequently documented on graphs known as titration curves, where the volume of the titrant is typically the independent variable, and the pH of the solution is the dependent variable (because it changes depending on the composition of the two solutions).