Chapter 2: Q10P (page 40)
Question: Draw the structures of the conjugate bases of the following acids:
a.
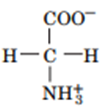
b.
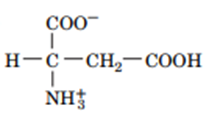
Short Answer
Answer
- The structure are:
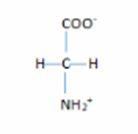
2. The structure are:
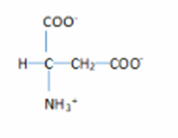
Learning Materials
Features
Discover
Chapter 2: Q10P (page 40)
Question: Draw the structures of the conjugate bases of the following acids:
a.

b.
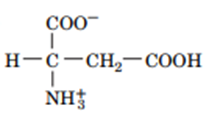
Answer

2. The structure are:

All the tools & learning materials you need for study success - in one app.
Get started for free
Describe how to calculate pH from the concentration of H+ or OH-.
Question: What must a buffer solution include to resist changes in pH by adding acid or base?
Where would the following substances partition in water containing palmitic acid micelles?
(a)
(b)
What is the relationship between polarity and hydrophobicity?
Question: (a) Would phosphoric acid or succinic acid be a better buffer at pH 5 ?
(b) Would ammonia or piperidine be a better buffer at pH 9 ?
(c) Would HEPES or Tris be a better buffer atpH7.5 ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.