Chapter 2: Q13CP (page 39)
Describe how to calculate pH from the concentration of H+ or OH-.
Short Answer
The pH can be calculated using pH formula.
Learning Materials
Features
Discover
Chapter 2: Q13CP (page 39)
Describe how to calculate pH from the concentration of H+ or OH-.
The pH can be calculated using pH formula.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Identify the potential hydrogen bond donors and acceptors in the following molecules:
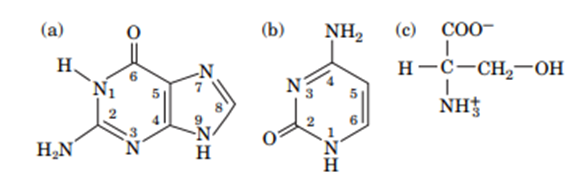
What is the relationship between polarity and hydrophobicity?
Calculate the pH of a 200 mL solution of pure water to which has been added 50 mL of 1 mM HCl.
Compare and contrast the strategies for eliminating waste nitrogen (as ammonia) and carbon dioxide in
(a) a terrestrial mammal,
(b) a freshwater fish, and
(c) a saltwater fish. Be sure to consider osmotic effects as well as acid-base balance.
Question: Explain why polar substances dissolve in water while nonpolar substances do not.
What do you think about this solution?
We value your feedback to improve our textbook solutions.