Chapter 2: Q16. (page 40)
What is the pK of the weak acid HA if a solution containing 0.1 M HA and 0.2 M A− has a pH of 6.5?
Short Answer
The pK value of the solution is 6.2.
Learning Materials
Features
Discover
Chapter 2: Q16. (page 40)
What is the pK of the weak acid HA if a solution containing 0.1 M HA and 0.2 M A− has a pH of 6.5?
The pK value of the solution is 6.2.
All the tools & learning materials you need for study success - in one app.
Get started for free
Sketch a diagram of a water molecule and indicate the ends that bear partial positive and negative charges.
What is the relationship between polarity and hydrophobicity?
Compare and contrast the strategies for eliminating waste nitrogen (as ammonia) and carbon dioxide in
(a) a terrestrial mammal,
(b) a freshwater fish, and
(c) a saltwater fish. Be sure to consider osmotic effects as well as acid-base balance.
Question: Draw the structures of the conjugate bases of the following acids:
a.
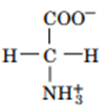
b.
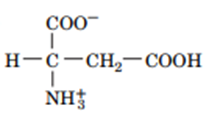
Question: Describe what happens when a dialysis bag containing pure water is suspended in a beaker of seawater. What would happen if the dialysis membrane were permeable to water but not solutes?
What do you think about this solution?
We value your feedback to improve our textbook solutions.