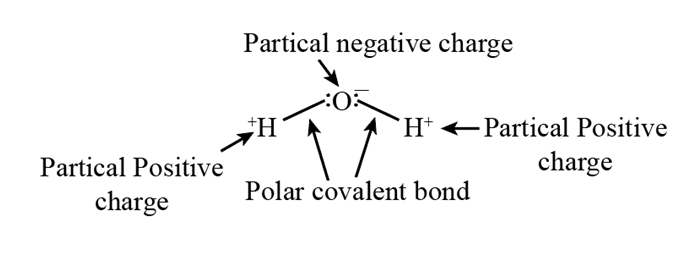
Chapter 2: 1CP (page 31)
Sketch a diagram of a water molecule and indicate the ends that bear partial positive and negative charges.
Short Answer
Diagram of a water molecule:
Learning Materials
Features
Discover
Chapter 2: 1CP (page 31)
Sketch a diagram of a water molecule and indicate the ends that bear partial positive and negative charges.
Diagram of a water molecule:

All the tools & learning materials you need for study success - in one app.
Get started for free
A solution is made by mixing 50 mL of 2.0 M K2HPO4 and 25 mL of 2.0 M KH2PO4. The solution is diluted to a final volume of 200 mL. What is the pH of the final solution?
Marble is composed largely of calcium carbonate (CaCO3). Spilling tomato juice on a slab of marble etches (eats away) the surface. Explain what happens in chemical terms.
An E. coli cell contains about 2.6 x 108ions, which constitute about 1% of the mass of the cell, and each ion has an average molecular mass of40g.
(a) What is the approximate mass of the cell?
(b) The same cell contains about 2 x 108 carbohydrate molecules, which have an average molecular mass of 150g. mol-1. Approximately what percentage of the cell's mass is carbohydrate?
(c) The molecular mass of E. coli DNA is about 5.6 x 109g . mol-1, and it accounts for about 6%of the cell's mass. How many DNA molecules does the cell contain?
Question: (a) Would phosphoric acid or succinic acid be a better buffer at pH 5 ?
(b) Would ammonia or piperidine be a better buffer at pH 9 ?
(c) Would HEPES or Tris be a better buffer atpH7.5 ?
Question: What must a buffer solution include to resist changes in pH by adding acid or base?
What do you think about this solution?
We value your feedback to improve our textbook solutions.