Chapter 19: Q. 43 (page 545)
A 150 L 1≈40 gal2 electric hot-water tank has a 5.0 kW heater.
How many minutes will it take to raise the water temperature from 65°F to 140°F?
Short Answer
The required time is 87mins
Learning Materials
Features
Discover
Chapter 19: Q. 43 (page 545)
A 150 L 1≈40 gal2 electric hot-water tank has a 5.0 kW heater.
How many minutes will it take to raise the water temperature from 65°F to 140°F?
The required time is 87mins
All the tools & learning materials you need for study success - in one app.
Get started for free
of nitrogen gas at and an initial pressure of undergo an isobaric expansion until the volume has tripled.
a. What are the gas volume and temperature after the expansion?
b. How much heat energy is transferred to the gas to cause this expansion?
The gas pressure is then decreased at constant volume until the original temperature is reached.
c. What is the gas pressure after the decrease?
d. What amount of heat energy is transferred from the gas as its pressure decreases?
e. Show the total process on a diagram. Provide an appropriate scale on both axes.
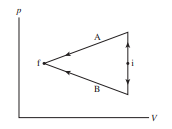
Shows two different processes taking an ideal gas from state i to state f.
Is the work done on the gas in process A greater than, less than, or equal to the work done in process B? Explain.
Two containers hold equal masses of nitrogen gas at equal temperatures. You supply 10 J of heat to container A while not allowing its volume to change, and you supply 10 J of heat to container B while not allowing its pressure to change. Afterward, is temperature TA greater than, less than, or equal to TB? Explain.
Draw a first-law bar chart (see Figure 19.12) for the gas process in
w
What do you think about this solution?
We value your feedback to improve our textbook solutions.