Chapter 29: Q. 64 (page 834)
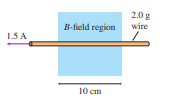
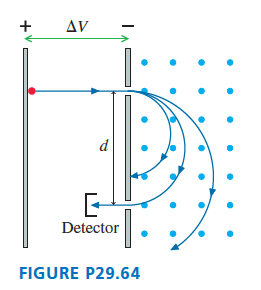
shows a mass spectrometer, an analytical instrument used to identify the various molecules in a sample by measuring their charge-to-mass ratio . The sample is ionized, the positive ions are accelerated (starting from rest) through a potential differencelocalid="1648976601527" , and they then enter a region of uniform magnetic field. The field bends the ions into circular trajectories, but after just half a circle they either strike the wall or pass through a small opening to a detector. As the accelerating voltage is slowly increased, different ions reach the detector and are measured. Consider a mass spectrometer with localid="1648976606181" a magnetic field and an localid="1648976610307" spacing between the entrance and exit holes. To five significant figures, what accelerating potential differences localid="1648978768434" are required to detect the ions localid="1648978902862" and localid="1648978898077" ? See Exercise localid="1648978753549" for atomic masses; the mass of the missing electron is less than localid="1648978758219" and is not relevant at this level of precision. Although localid="1648978910549" and localid="1648978778238" both have a nominal molecular mass of localid="1648978782098" , they are easily distinguished by virtue of their slightly different accelerating voltages. Use the following constants:
Short Answer
Part
For,the potential difference is
Part
For ,the potential difference is
Part
For,the potential difference is