Chapter 37: Q. 39 (page 1083)
The diameter of an atom is and the diameter of its nucleus is. What percent of the atom's volume is occupied by mass and what percent is empty space?
Short Answer
The volume occupied by mass
Learning Materials
Features
Discover
Chapter 37: Q. 39 (page 1083)
The diameter of an atom is and the diameter of its nucleus is. What percent of the atom's volume is occupied by mass and what percent is empty space?
The volume occupied by mass
All the tools & learning materials you need for study success - in one app.
Get started for free
Identify the element, the isotope, and the charge state of each atom in FIGURE . Give your answer in symbolic form, such as 4He+ or 8Be-.
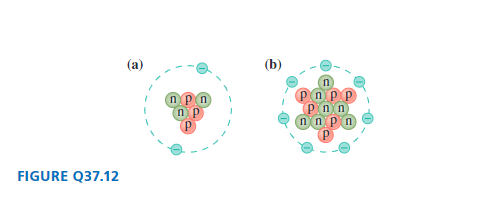
Figure 37.7 identified the wavelengths of four lines in the Balmer series of hydrogen.
a. Determine the Balmer formula n and m values for these wavelengths.
b. Predict the wavelength of the fifth line in the spectrum .
10. Explain why the observation of alpha particles scattered at very large angles led Rutherford to reject Thomson’s model of the atom and to propose a nuclear model.
Explain why the observation of alpha particles scattered at very large angles led Rutherford to reject Thomson’s model of the atom and to propose a nuclear model.
What do you think about this solution?
We value your feedback to improve our textbook solutions.