Chapter 37: Q. 3 - Excercises And Problems (page 1081)
The wavelengths in the hydrogen spectrum with m=1 form a series of spectral lines called the Lyman series. Calculate the wavelengths of the first four members of the Lyman series
Learning Materials
Features
Discover
Chapter 37: Q. 3 - Excercises And Problems (page 1081)
The wavelengths in the hydrogen spectrum with m=1 form a series of spectral lines called the Lyman series. Calculate the wavelengths of the first four members of the Lyman series
All the tools & learning materials you need for study success - in one app.
Get started for free
An alpha particle (a bare helium nucleus with (q=+2e) accelerates across a 100 V potential difference, starting from rest. What is the particle’s kinetic energy in eV when it reaches the negative electrode? This question requires no mathematics beyond what you can do in your head.
A 222Rn atom (radon) in a 0.75 T magnetic field undergoes radioactive decay, emitting an alpha particle in a direction perpendicular to . The alpha particle begins cyclotron motion with a radius of 45 cm. With what energy, in MeV, was the alpha particle emitted?
Electrons pass through the parallel electrodes shown in FIGURE EX37.9 with a speed of . What magnetic field strength and direction will allow the electrons to pass through without being deflected? Assume that the magnetic field is confined to the region between the electrodes.
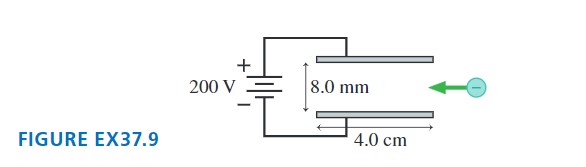
Figure 37.7 identified the wavelengths of four lines in the Balmer series of hydrogen.
(a) Determine the Balmer formula n and m values for these wavelengths.
(b) Predict the wavelength of the fifth line in the spectrum

What do you think about this solution?
We value your feedback to improve our textbook solutions.