Chapter 38: Q. 35 (page 1115)
What is the third-longest wavelength in the absorption spectrum of hydrogen?
Short Answer
The wavelength of the third longest wave is 97.3 nm
Learning Materials
Features
Discover
Chapter 38: Q. 35 (page 1115)
What is the third-longest wavelength in the absorption spectrum of hydrogen?
The wavelength of the third longest wave is 97.3 nm
All the tools & learning materials you need for study success - in one app.
Get started for free
dbuhyhyerbguybsuyrg
The diameter of the nucleus is about . What is the kinetic energy, in MeV, of a proton with a de Broglie wavelength of ?
The first three energy levels of the fictitious element X are shown in Figure.
a. What is the ionization energy of element X?
b. What wavelengths are observed in the absorption spectrum of element X? Express your answers in nm.
c. State whether each of your wavelengths in part b corresponds to ultraviolet, visible, or infrared light.
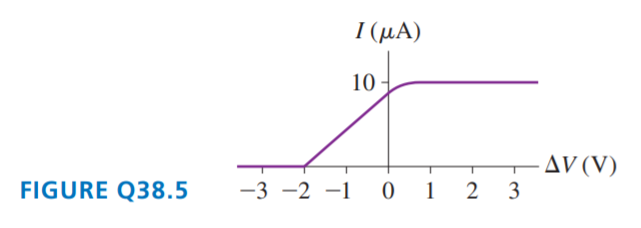
FIGURE Q38.5 is the current-versus-potential-difference graph for a photoelectric-effect experiment with an unknown metal. If classical physics provided the correct description of the photoelectric effect, how would the graph look if:
a. The light was replaced by an equally intense light with a shorter wavelength? Draw it.
b. The metal was replaced by a different metal with a smaller work function? Draw it.
How would the graphs of Figure 38.3 look if classical physics provided the correct description of the photoelectric effect? Draw the graph and explain your reasoning. Include curves for both weak light and intense light
What do you think about this solution?
We value your feedback to improve our textbook solutions.