Chapter 40: Q. 1 (page 1174)
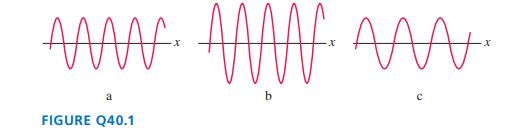
FIGURE Qshows the de Broglie waves of three equal-mass particles. Which one is moving most slowly? Explain.
Short Answer
The wave is shown in part (c) one is moving most slowly.
Learning Materials
Features
Discover
Chapter 40: Q. 1 (page 1174)
FIGURE Qshows the de Broglie waves of three equal-mass particles. Which one is moving most slowly? Explain.

The wave is shown in part (c) one is moving most slowly.
All the tools & learning materials you need for study success - in one app.
Get started for free
Consider a particle in a rigid box of length L. For each of the states and :
a. Sketch graphs of . Label the points and .
b. Where, in terms of L, are the positions at which the particle is most likely to be found?
c. Where, in terms of L, are the positions at which the particle is least likely to be found?
d. Determine, by examining your graphs, if the probability of finding the particle in the left one-third of the box is less than, equal to, or greater than . Explain your reasoning.
e. Calculate the probability that the particle will be found in the left one-third of the box
A -long box has a thin partition that divides the box into a-long section and a-long section. An electron confined in the shorter section is in the state. The partition is briefly withdrawn, then reinserted, leaving the electron in the longer section of the box. What is the electron’s quantum state after the partition is back in place?
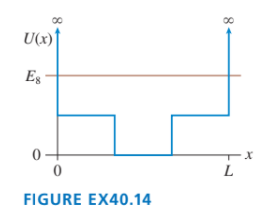
Sketch the wave function for the potential energy shown in FIGURE EX40.14.
The energy of an electron in a deep potential well is . At what distance into the classically forbidden region has the amplitude of the wave function decreased to of its value at the edge of the potential well?
A typical electron in a piece of metallic sodium has energycompared to a free electron, where is the work function of sodium.
a. At what distance beyond the surface of the metal is the electron’s probability density of its value at the surface?
b. How does this distance compare to the size of an atom?
What do you think about this solution?
We value your feedback to improve our textbook solutions.