Chapter 21: Q. 46 (page 570)
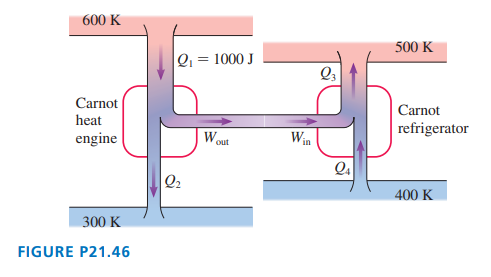
shows a Carnot heat engine driving a Carnot refrigerator.
a. Determine and .
b. Is greater than, less than, or equal to?
c. Do these two devices, when operated together in this way, violate the second law?
Short Answer
a. .
b. is greater than .
c. No, the two devices don't violet second law of thermodynamics.