Chapter 21: Q. 18 (page 595)
At what pressure ratio does a Brayton cycle using a monatomic gas have an efficiency of ?
Short Answer
The pressure ratio using a monatomic gas,
Learning Materials
Features
Discover
Chapter 21: Q. 18 (page 595)
At what pressure ratio does a Brayton cycle using a monatomic gas have an efficiency of ?
The pressure ratio using a monatomic gas,
All the tools & learning materials you need for study success - in one app.
Get started for free
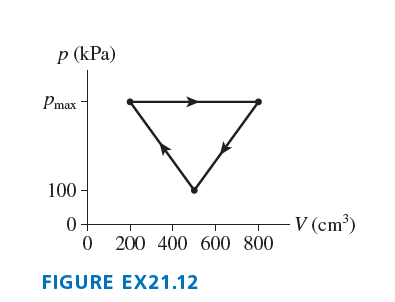
A gas following the trajectory of FIGURE does of work per cycle. What is ?
Consider a power plant (this is the useful output in the form of electric energy) that operates between 30oC and 450oC at 65% of the Carnot efficiency. This is enough electric energy for about 750 homes. One way to use energy more efficiently would be to use the 30oC“waste” energy to heat the homes rather than releasing that heat energy into the environment. This is called cogeneration, and it is used in some parts of Europe but rarely in the United States. The average home uses 70GJof energy per year for heating. For estimating purposes, assume that all the power plant’s exhaust energy can be transported to homes without loss and that home heating takes place at a steady rate for half a year each year. How many homes could be heated by the power plant?
A heat engine using a diatomic ideal gas goes through the following closed cycle:
Isochoric cooling until the pressure is restored to its initial value. What are the thermal efficiencies of () this heat engine and
() a Carnot engine operating between the highest and lowest temperatures reached by this engine?
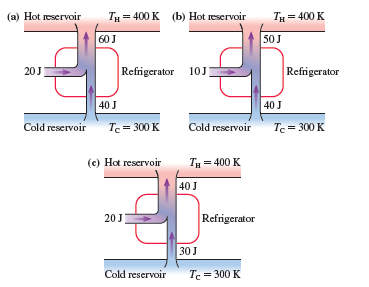
Which, if any, of the refrigerators in FIGURE EX21.23 violate (a) the first law of thermodynamics or (b) the second law of thermodynamics? Explain.
The engine that powers a crane burns fuel at a flame temperature of 2000°C. It is cooled by 20°C air. The crane lifts a 2000 kg steel girder 30 m upward. How much heat energy is transferred to the engine by burning fuel if the engine is 40% as efficient as a Carnot engine?
What do you think about this solution?
We value your feedback to improve our textbook solutions.