Chapter 21: Heat Engines and Refrigerators
Q. 49
A car’s internal combustion engine can be modeled as a heat engine operating between a combustion temperature of 1500oC and an air temperature of 20oC with 30% of the Carnot efficiency. The heat of combustion of gasoline is 47kJ/g. What mass of gasoline is burned to accelerate a 1500kgcar from rest to a speed of 30 m/s?
Q. 5
Rank in order, from largest to smallest, the thermal efficiencies to of the four heat engines in FIGURE Q21.5. Explain.
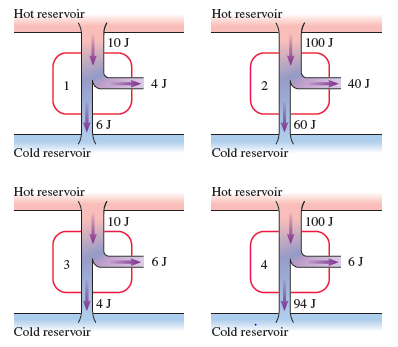
Q. 5
A refrigerator requires of work and exhausts of heat per cycle. What is the refrigerator’s coefficient of performance?
Q. 50
Consider a power plant (this is the useful output in the form of electric energy) that operates between 30oC and 450oC at 65% of the Carnot efficiency. This is enough electric energy for about 750 homes. One way to use energy more efficiently would be to use the 30oC“waste” energy to heat the homes rather than releasing that heat energy into the environment. This is called cogeneration, and it is used in some parts of Europe but rarely in the United States. The average home uses 70GJof energy per year for heating. For estimating purposes, assume that all the power plant’s exhaust energy can be transported to homes without loss and that home heating takes place at a steady rate for half a year each year. How many homes could be heated by the power plant?
Q. 51
A typical coal-fired power plant burns metric tons of coal every hour to generate of electricity. metric ton = . The density of coal is 1500 kg/m3 and its heat of combustion is . Assume that all heat is transferred from the fuel to the boiler and that all the work done in spinning the turbine is transformed into electric energy.
a. Suppose the coal is piled up in a room. How tall must the pile be to operate the plant for one day?
b. What is the power plant’s thermal efficiency?
Q. 52
A nuclear power plant generates of heat energy from nuclear reactions in the reactor’s core. This energy is used to boil water and produce high-pressure steam at . The steam spins a turbine, which produces of electric power, then the steam is condensed and the water is cooled to before starting the cycle again.
a. What is the maximum possible thermal efficiency of the power plant?
b. What is the plant’s actual efficiency?
c. Cooling water from a river flows through the condenser (the low-temperature heat exchanger) at the rate of ( million gallons per hour). If the river water enters the condenser at , what is its exit temperature?
Q. 53
The electric output of a power plant is . Cooling water flows through the power plant at the rate . The cooling water enters the plant at and exits at. What is the power plant’s thermal efficiency?
Q. 54
Engineers testing the efficiency of an electric generator gradually vary the temperature of the hot steam used to power it while leaving the temperature of the cooling water at a constant . They find that the generator’s efficiency increases at a rate at steam temperatures near . What is the ratio of the generator’s efficiency to the efficiency of a Carnot engine?
Q. 55
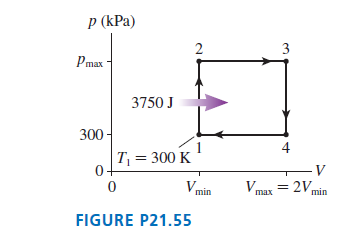
A heat engine using of a monatomic gas follows the cycle shown in FIGURE P. of heat energy is transferred to the gas during process .
a. Determine , and for each of the four processes in this cycle. Display your results in a table.
b. What is the thermal efficiency of this heat engine?
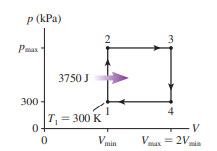
Q. 55
A heat engine using 1.0 mol of a monatomic gas follows the cycle shown in FIGURE P21.55. 3750 J of heat energy is transferred to the gas during process 1 -> 2.
a. Determine Ws, Q, and ΔEth for each of the four processes in this cycle. Display your results in a table.
b. What is the thermal efficiency of this heat engine?